| CPC A61K 47/643 (2017.08) [A61K 31/337 (2013.01); A61K 31/4745 (2013.01); A61P 15/00 (2018.01); A61P 35/00 (2018.01)] | 14 Claims |
|
1. A method of treating cancer in a patient suffering from platinum-resistant ovarian, fallopian tube, uterine, cervical, vaginal, vulvar, or peritoneal cancer and having a cancerous tumor that expresses high levels of mRNA encoding the glucocorticoid receptor gene NR3C1, comprising:
a) determining that the expression level of mRNA encoding the glucocorticoid receptor gene NR3C1 in a sample of said cancerous tumor is higher than the median level of mRNA expression in said sample of the cancerous tumor, wherein
i) said cancerous tumor is selected form the group consisting of ovarian, fallopian tube, uterine, cervical, vaginal, vulvar, and peritoneal cancer;
ii) said patient suffered from progression of said cancer within six months after receiving a platinum-based cancer chemotherapy treatment; and
iii) the patient has received at least one prior systemic cancer treatment comprising administration of bevacizumab; and
b) administering to the patient a cancer chemotherapy agent comprising a taxane,
wherein said taxane cancer chemotherapy agent is selected from the group of taxane cancer chemotherapy agents consisting of paclitaxel, nab-paclitaxel, docetaxel, larotaxel, tesetaxel, cabazitaxel, and ortataxel,
said taxane administration comprising administration according to a cancer chemotherapy dosing schedule,
said cancer chemotherapy dosing schedule requiring that there be at least one day without administration of said taxane cancer chemotherapy agent between days on which the taxane cancer chemotherapy agent is administered to the patient;
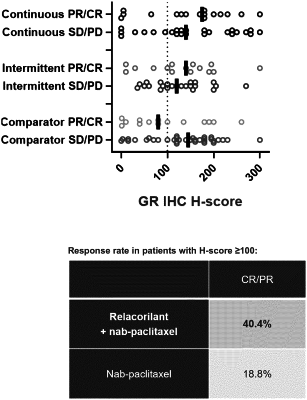
c) intermittently administering to the patient an effective amount of the heteroaryl ketone fused azadecalin glucocorticoid receptor modulator compound (R)-(1-(4-fluorophenyl)-6-((1-methyl-1H-pyrazol-4-yl)sulfonyl)-4,4a,5,6,7,8-hexahydro-1H-pyrazolo[3,4-g]isoquinolin-4a-yl)(4-(trifluoromethyl)pyridin-2-yl)methanone (“relacorilant”), which has the following structure:
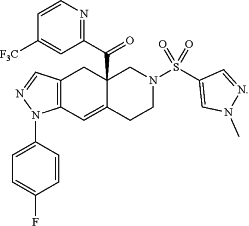 wherein said intermittent administration comprises administration of said relacorilant on the day before, the day of, and the day after said taxane cancer chemotherapy agent is administered to the patient,
thereby treating said platinum-resistant center in the patient i) identified as hosting a cancerous tumor that expresses high levels of mRNA encoding the glucocorticoid receptor gene NR3C1 as compared to the median level of mRNA expression in the cancerous tumor, ii) who suffered from disease progression within six months after receiving a platinum-based cancer chemotherapy treatment, and iii) who has received at least one prior systemic cancer treatment comprising administration of bevacizumab.
|