| CPC A61K 31/675 (2013.01) [A61K 31/416 (2013.01); A61K 31/4439 (2013.01); A61K 31/497 (2013.01); A61K 45/06 (2013.01)] | 20 Claims |
|
1. A pharmaceutical composition, comprising a compound selected from compounds of Formula Ia and pharmaceutically acceptable salts, solvates, hydrates, and N-oxides thereof:
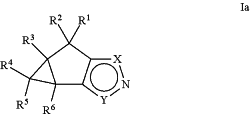 wherein:
R1, R2, R3, R4, R5, and R6 are each independently selected from: H and C1-C6 alkyl;
X is NR7 and Y is CC(O) N(R8) R9; or
X is CC(O) N(R8) R9 and Y is NR7;
R7 is —R10—R11—R12-R13, wherein:
R10 is selected from: C1-C6 alkylene, heteroarylene, and heterocyclylene; or R10 is absent;
R11 is selected from: —C(O) NH— and C1-C6 alkylene; or R11 is absent;
R12 is C1-C6 alkylene; or R12 is absent; and
R13 is selected from: C1-C6 alkyl, aryl, C3-C7 cycloalkyl, heteroaryl, heterocyclyl, and hydroxyl; wherein said C1-C6 alkyl, aryl, and heteroaryl are each optionally substituted with one or two substituents selected from: C1-C6 alkoxy, C1-C6 alkyl, C1-C6 alkylamino, C1-C6 alkylsulfonyl, amino, C3-C7 cycloalkyl, cyano, C2-C8 dialkylamino, C1-C6 haloalkyl, halogen, and hydroxyl;
R8 is —R14—R15—R16—R17, wherein:
R14 is selected from: C1-C6 alkylene, C3-C7 cycloalkenylene, C3-C7 cycloalkylene, heteroarylene, and heterocyclylene; wherein said C1-C6 alkylene and heterocyclylene are each optionally substituted with one or more substituents selected from: C1-C6 alkoxycarbonyl, C1-C6 alkyl, C3-C7 cycloalkyl, aryl, carboxy, heteroaryl, heterocyclyl, and hydroxyl; wherein said C1-C6 alkyl and aryl are optionally substituted with one substituent selected from: C1-C6 alkoxy, aryl, halogen, heteroaryl, and hydroxyl; or R14 is absent;
R15 is selected from: —C(O) NH—,—C(O)—,—C(O)O—, C1-C6 alkylene, C3-C7 cycloalkylene, heteroarylene, and heterocyclylene; wherein said heterocyclylene is optionally substituted with C1-C6 alkyl; or R15 is absent;
R16 is C1-C6 alkylene; or R16 is absent; and
R17 is selected from: H, C1-C6 alkoxy, C1-C6 alkyl, C1-C6 alkylamino, C1-C6 alkylcarboxamide, C2-C6 alkynyl, ureyl, amino, aryl, arylamino, arylcarbonyl, aryloxy, carbo-C1-C6-alkoxy, carboxamide, carboxy, cyano, C3-C7 cycloalkyl, C5-C11 bicycloalkyl, C3-C7 cycloalkylamino, C2-C8 dialkylamino, C2-C8 dialkylsulfonamide, C1-C6 haloalkyl, heteroaryl, heteroaryloxy, heterobicyclyl, heterocyclyl, hydroxyl, and phosphonooxy; wherein said C1-C6 alkylamino, amino, aryl, arylamino, aryloxy, C5-C11 bicycloalkyl, C3-C7 cycloalkyl, C3-C7 cycloalkylamino, heteroaryl, heterobicyclyl, heterocyclyl, and ureyl are each optionally substituted with one or more substituents selected from: C1-C6 alkoxy, C1-C6 alkoxycarbonyl, C1-C6 alkyl, C1-C6 alkylsulfonyl, amino, aryl, carboxy, cyano, C3-C7 cycloalkyl, C2-C8 dialkylamino, C1-C6 haloalkoxy, C1-C6 haloalkyl, halogen, heteroaryl, heterocyclyl, and hydroxyl; and
R9 is selected from: H, C1-C6 alkyl, and C3-C7 cycloalkyl; or
R8 and R9 together with the nitrogen atom to which they are both bonded form a group selected from: heterocyclyl and heterobicyclyl, each optionally substituted with one or more substituents selected from: Carbo-C1-C6-alkoxy, C1-C6 alkoxy, C1-C6 alkyl, aryl, carbo-C1-C6-alkoxy, C1-C6 haloalkyl, halogen, heteroaryl, heteroaryloxy, heterocyclyl, and hydroxyl; wherein said aryl, C1-C6 alkyl, and heteroaryl are optionally substituted with one substituent selected from: C3-C7 cycloalkyl, C1-C6 alkoxy, halogen, and hydroxyl,
and one or more anticancer agents selected from the group consisting of acivicin, aclarubicin, acodazole hydrochloride, acronine, adozelesin, aldesleukin, altretamine, ambomycin, ametantrone acetate, aminoglutethimide, amsacrine, anthramycin, asparaginase, asperlin, 4zacytidine, azetepa, azotomycin, batimastat, benzodepa, bicalutamide, bisantrene hydrochloride, bisnafide dimesylate, bizelesin, bleomycin sulfate, brequinar sodium, bropirimine, busulfan, cactinomycin, calusterone, caracemide, carbetimer, carboplatin, carmustine, carubicin hydrochloride, carzelesin, cedefingol, chlorambucil, cirolemycin, cisplatin, cladribine, crisnatol mesylate, cyclophosphamide, cytarabine, dacarbazine, dactinomycin, daunorubicin hydrochloride, decitabine, dexormaplatin, dezaguanine, dezaguanine mesylate, diaziquone, doxorubicin, doxorubicin hydrochloride, droloxifene, droloxifene citrate, dromostanolone propionate, duazomycin, edatrexate, eflornithine hydrochloride, elsamitrucin, enloplatin, enpromate, epipropidine, epirubicin hydrochloride, erbulozole, esorubicin hydrochloride, estramustine, estramustine phosphate sodium, etanidazole, etoposide, etoposide phosphate, etoprine, fadrozole hydrochloride, fazarabine, fenretinide, floxuridine, fludarabine phosphate, fluorouracil, fluorocitabine, fosquidone, fostriecin sodium, gemcitabine, hydroxyurea, idarubicin hydrochloride, ifosfamide, ilmofosine, interleukin II, recombinant interleukin II, interferon alfa-2a, interferon alfa-2b, interferon alfa-n1, interferon alfa-n3, interferon beta-I a, interferon gamma-I b, iproplatin, irinotecan hydrochloride, lanreotide acetate, liarozole hydrochloride, lometrexol sodium, lomustine, losoxantrone hydrochloride, masoprocol, maytansine, mechlorethamine hydrochloride, megestrol acetate, melengestrol acetate, melphalan, menogaril, mercaptopurine, methotrexate, methotrexate sodium, metoprine, meturedepa, mitindomide, mitocarcin, mitocromin, mitogillin, mitomalcin, mitomycin, mitosper, mitotane, mitoxantrone hydrochloride, mycophenolic acid, nocodazole, nogalamycin, ormaplatin, oxisuran, paclitaxel, pegaspargase, peliomycin, pentamustine, peplomycin sulfate, perfosfamide, pipobroman, piposulfan, piroxantrone hydrochloride, plicamycin, plomestane, porfimer sodium, porfiromycin, prednimustine, procarbazine hydrochloride, puromycin, puromycin hydrochloride, pyrazofurin, riboprine, rogletimide, safingol, safingol hydrochloride, semustine, simtrazene, sparfosate sodium, sparsomycin, spirogermanium hydrochloride, spiromustine, spiroplatin, streptonigrin, streptozocin, sulofenur, talisomycin, tecogalan sodium, tegafur, teloxantrone hydrochloride, temoporfin, teniposide, teroxirone, testolactone, thiamiprine, thioguanine, thiotepa, tiazofurin, tirapazamine, toremifene citrate, trestolone acetate, triciribine phosphate, trimetrexate, trimetrexate glucuronate, tubulozole hydrochloride, uracil mustard, uredepa, vapreotide, verteporfin, vinblastine sulfate, vincristine sulfate, vindesine, vindesine sulfate, vinepidine sulfate, vinglycinate sulfate, vinleurosine sulfate, vinorelbine tartrate, vinrosidine sulfate, vinzolidine sulfate, vorozole, zeniplatin, zinostatin, and zorubicin hydrochloride; in an amount that is therapeutically effective in treating cancer in an individual, wherein the cancer is not prostate cancer, leukemia, lymphoma, or a CNS tumor.
|