| CPC C25D 3/562 (2013.01) [C25D 5/18 (2013.01); C25D 5/627 (2020.08); C25D 5/611 (2020.08)] | 20 Claims |
|
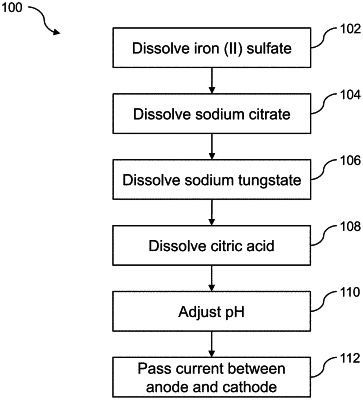
1. A method for plating a substrate using an electrolyte solution, the method comprising:
preparing the electrolyte solution consisting of an aqueous medium; a divalent iron salt consisting of iron (II) sulfate, iron (II) halide, iron (II) nitrate, iron (II) acetate, or iron (II) perchlorate; an alkali metal citrate; a tungstate salt consisting of an alkali metal tungstate; a citric acid; and sodium hydroxide, potassium hydroxide, sulfuric acid, or a combination thereof by:
dissolving in the aqueous medium, the divalent iron salt in an amount ranging from about 0.05 to about 0.5 mol per liter of the electrolyte solution and the alkali metal citrate in an amount ranging from about 0.05 to about 2 mol per liter of the electrolyte solution to form a first solution;
dissolving in the first solution the tungstate salt in an amount ranging from about 0.1 to about 1.5 mol per liter of the electrolyte solution to form a second solution; and
dissolving in the second solution the citric acid in an amount ranging from about 0.01 to about 1 mol per liter of the electrolyte solution to form the electrolyte solution;
passing a current between a cathode and an anode through the electrolyte solution to deposit iron and tungsten on the substrate; and
forming a coating layer comprising an alloy of iron and tungsten when the pH of the electrolyte solution is maintained at about 3 to about 12.
|