| CPC G01N 33/5047 (2013.01) [C07K 16/2818 (2013.01); G01N 33/57492 (2013.01); C12N 5/0634 (2013.01); C12N 2533/54 (2013.01); C12N 2533/76 (2013.01); G01N 33/5091 (2013.01); G01N 2800/245 (2013.01); G01N 2800/52 (2013.01)] | 13 Claims |
|
1. A method to quantitatively measure the response of a patient to an immune-modulator drug, the method comprising:
a) activating peripheral blood mononuclear cells (PBMCs), total leukocytes or specific sub-populations of PBMCs, obtained from a biological sample selected from the group consisting of blood and a blood cellular derivative of the patient, either: i) though incubation with a lymphocyte activation compound selected from the group consisting of agonistic antibodies anti-CD3 (T-cell Receptor (TCR)) and anti-CD28, ionomycin and PMA (phorbol myristate acetate), a lectin, a superantigen, and a Lipopolysaccharide (LPS); or ii) through a Mix Lymphocyte Reaction, to obtain activated PBMCs;
b) obtaining a hydrogel comprising the activated PBMCs, total leukocytes or specific sub-populations of PBMCs, embedded within the hydrogel by incorporating the activated cells into a hydrogel, wherein;
the hydrogel is located in a support comprising channelled wells having dimensional proportions in which the longitudinal axis of the spatial three axes is at least 4 times longer than the length average of the two other axes, so that the hydrogel is capable of providing a measurable and stable drug gradient flux in the longitudinal axis of channelled wells, and
the channelled wells contain a hydrogel volume per channel of at least 40 μl;
c) contacting the hydrogel of step b) with one or more immune-modulator drugs;
d) adding a solution comprising a compound capable of providing an absorbance, fluorescence or luminescence signal, thereby defining an inhibitory zone around a site of contact of an immunosuppressant drug with the hydrogel; and
e) obtaining a quantification of the immune-modulator drug gradient formed, wherein the quantification is obtained by image acquisition or quantification of the absorbance, fluorescence or luminescence signal,
wherein:
the hydrogel is formed through non-covalent crosslinking of polymer chains, wherein the total polymeric fraction represents less than 5% (5 gr/100 ml of hydrogel);
the hydrogel composition generates a non-toxic environment able to sustain cell proliferation having adequate nutritional composition and stiffness, and absent of unspecific induction of PBMCs activation; and
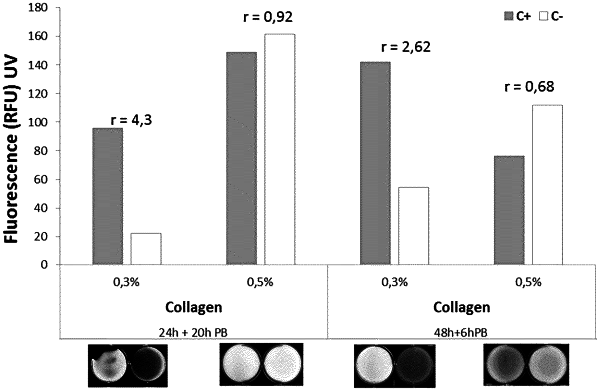
the total polymeric fraction of the hydrogel consists of collagen, wherein the collagen content is present at below or equal to 0.3% (gr/100 ml of hydrogel).
|