| CPC G01N 33/15 (2013.01) [B01D 63/087 (2013.01); B01D 69/10 (2013.01); B01D 2313/025 (2013.01); B01D 2325/0283 (2022.08)] | 16 Claims |
|
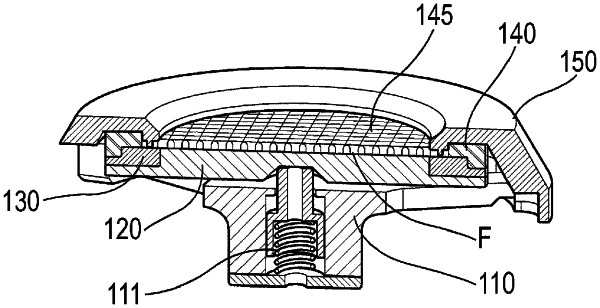
1. An in vitro release testing (IVRT) device for orally inhaled drug products, for use in an IVRT apparatus, said device comprising an air-permeable filter loaded with particulate material representing a dose of an orally inhaled drug product, said device comprising:
an upper filter support element and a lower filter support element, said loaded filter being circumferentially retained between said upper and lower support elements,
a filter cover to cover the upper surface of said loaded filter, and
a filter cover retainer provided to assemble and seal the IVRT device, said filter cover retainer having a spring to control the compressive load between said lower filter support element, said filter, said upper filter support element and said filter cover.
|