| CPC C07D 471/20 (2013.01) [A61K 31/438 (2013.01); A61P 31/14 (2018.01); C07D 495/04 (2013.01)] | 11 Claims |
|
1. A method of treating dengue disease comprising administering a Dengue NS4B specific inhibitor to a subject having dengue disease, wherein the Dengue NS4B specific inhibitor is a compound chosen from:
(a) a compound of Formula (I), wherein:
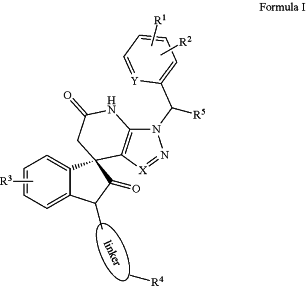 wherein:
R1, R2 are independently chosen from H, F, Cl, Br and CF3;
R3 is F, Cl or Br;
R4 is F, Cl, Br, alkyl, alkenyl, alkynyl, alkoxy, hydroxyl, carboxyl, ester, acetal, amino, 5-membered cycle or heterocycle, or 6-membered cycle or heterocycle;
R5 is H or alkyl;
X, Y are independently chosen from CH and N; and
linker is a 1-20 atom length carbon chain or, wherein said chain optionally includes one or more ester bonds, amide bonds or oxygen atoms; or
(b) a compound of Formula (Ib), wherein:
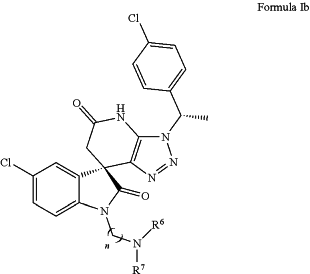 wherein R6, R7 are independently chosen from H and alkyl; or R6, R7 taken together with the N atom to which they are attached to form 5-membered or 6-membered heterocycle rings; and
n is 1-10; or
(c) a compound of formula (Ic), wherein:
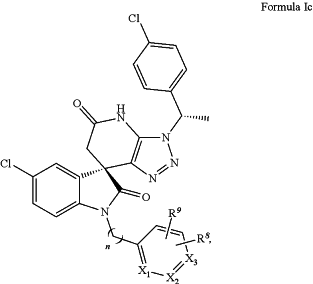 wherein R8, R9 are independently chosen from H, alkyl, alkoxy, F, Cl, Br, CF3, hydroxyl, carboxyl, ester, cyano, amino, and nitro, or R8, R9 taken together with the atoms to which they are attached form 5-membered or 6-membered fused ring;
X1, X2, X3 are independently chosen from CH and N; and
n is 1-10; or
(d) a compound represented by:
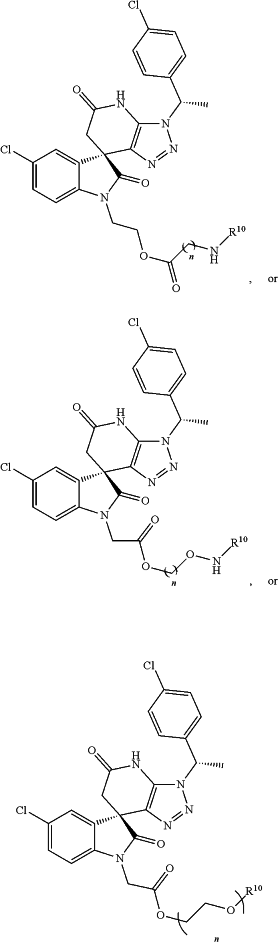 wherein R10 is chosen from
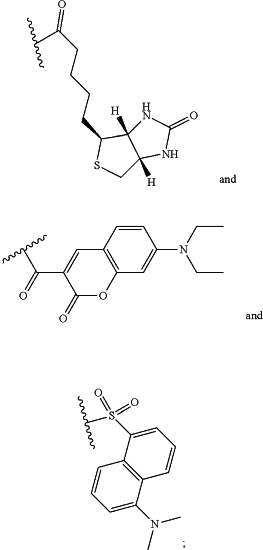 and
n is 1-10; or
(e) a compound represented by:
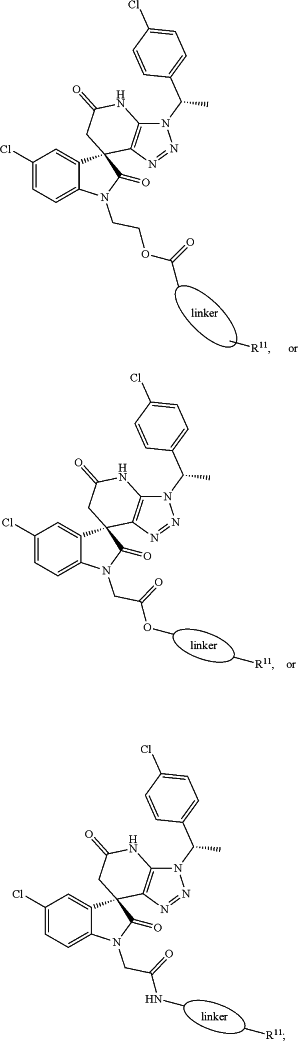 wherein R11 is chosen from
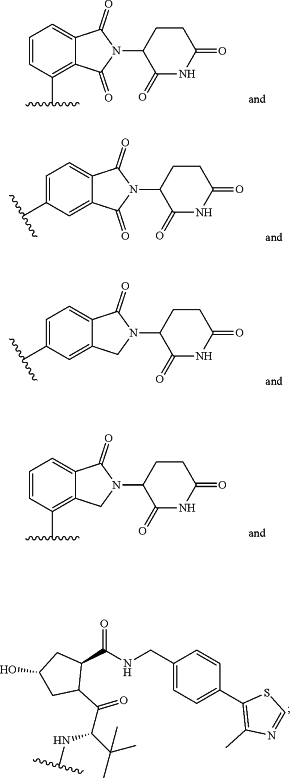 wherein linker is a 1-20 atom length carbon chain, wherein said chain optionally includes one or more ester bonds, amide bonds or oxygen atoms; or
(f) a compound of Formula (II)
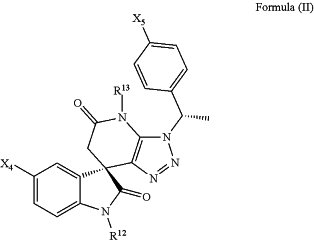 wherein X4, X5 are independently chosen from F, Cl, Br, and CF3;
R12 is a 1-20 atom length carbon chain; wherein said chain is optionally substituted with one or more ester bonds, amide bonds or oxygen atoms; wherein said chain is tethered to a terminal group chosen from F, Cl, Br, alkyl, alkenyl, alkynyl, alkoxy, hydroxyl, carboxyl, ester, acetal, amino, 5-membered cycle or heterocycle, or 6-membered cycle and heterocycle; and
R13 is H.
|