| CPC C07D 213/55 (2013.01) [C07D 213/79 (2013.01); C07D 213/89 (2013.01); C07D 401/04 (2013.01); C07D 471/04 (2013.01); C09K 8/03 (2013.01); C09K 11/07 (2013.01); E21B 43/16 (2013.01); E21B 47/11 (2020.05); E21B 49/08 (2013.01); G01N 21/6408 (2013.01); G01N 21/643 (2013.01); C09K 2211/1018 (2013.01); C09K 2211/182 (2013.01); E21B 49/0875 (2020.05); G01N 2021/641 (2013.01); G01N 2201/129 (2013.01)] | 18 Claims |
|
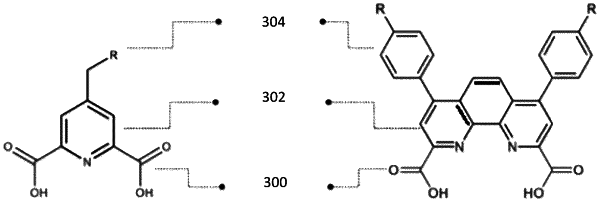
1. A complexing agent of Formula (I), or an anion or salt thereof:
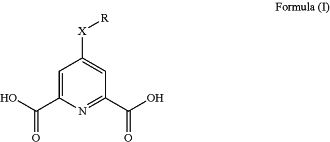 wherein:
X is present or absent, and when present, is selected from the group consisting of: C1-10 alkylene, C2-10 alkenylene, and C2-10 alkynylene, wherein each C1-10 alkylene, C2-10 alkenylene, and C2-10 alkynylene is optionally interrupted by one O, S, or NH;
R is -heteroaryl including from 5-10 ring atoms, wherein from 1-4 ring atoms are each independently selected from the group consisting of N, NH, O, and S, wherein the heteroaryl is optionally substituted with from 1-3 Rb;
each occurrence of Rb is independently selected from the group consisting of:
(i) halo;
(ii) cyano;
(iii) C1-6 alkyl;
(iv) C2-6 alkenyl;
(v) C2-6 alkynyl;
(vi) C1-4 haloalkyl;
(vii) C1-4 alkoxy;
(viii) C1-4 haloalkoxy;
(ix) —(C0-3 alkylene)—C3-6 cycloalkyl optionally substituted with from 1-4 independently selected C1-4 alkyl;
(x) —(C0-3 alkylene)-heterocyclyl including from 3-10 ring atoms, wherein from 1-3 ring atoms are each independently selected from the group consisting of NH, O, and S, wherein the heterocyclyl is optionally substituted with from 1-4 independently selected C1-4 alkyl;
(xi) —(C0-3 alkylene)-phenyl;
(xii) —(C0-3 alkylene)-heteroaryl including from 5-10 ring atoms, wherein from 1-4 ring atoms are each independently selected from the group consisting of N, NH, O, and S;
(xiii) —S(O)1-2(C1-4 alkyl); and
(xiv) —NR′R″;
(xv) —OH;
(xvi) —S(O)1-2(NR′R″);
(xvii) C1-4 thioalkoxy;
(xviii) —NO2;
(xix) —N(R′)(C(═O)C1-3 alkyl);
(xx) —C(═O)(C1-4 alkyl);
(xxi) —C(═O)O(C1-4 alkyl);
(xxii) —C(═O)OH, and
(xxiii) —C(═O)N(R′)(R″),
(xxiv) —(C1-6 alkyl)SO3; and
each occurrence of R′ and R″ is independently selected from the group consisting of: H and C1-4 alkyl; or, if R′ and R″ are bonded to the same atom, R′ and R″ together with the atom to which each is attached forms a ring including from 3-8 ring atoms, wherein the ring includes:
(a) from 1-7 ring carbon atoms; and (b) from 0-3 ring heteroatoms (in addition to the atom attached to R′ and R″), which are each independently selected from the group consisting of N, NH, O, and S.
|