| CPC A61K 47/549 (2017.08) [A61K 47/62 (2017.08); C07H 5/06 (2013.01); C07H 7/02 (2013.01); C07H 9/02 (2013.01); C07H 9/04 (2013.01); C07H 15/203 (2013.01); C07H 17/00 (2013.01); C07H 17/02 (2013.01); C07H 19/02 (2013.01); C07H 19/044 (2013.01); H03L 7/0814 (2013.01); H03L 7/0818 (2013.01); H03L 7/0998 (2013.01)] | 26 Claims |
|
1. A compound of the formula:
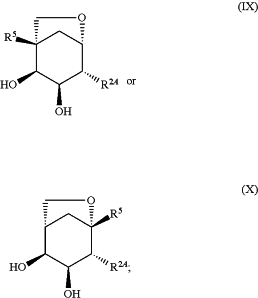 or a pharmaceutically acceptable salt thereof,
wherein:
R2A is selected from —NR6-heteroaryl and —NR6-aryl each of which aryl or heteroaryl is optionally substituted with 1, 2, 3, or 4 substituents independently selected from the group consisting of alkyl, haloalkyl, —OR6, F, Cl, Br, —NR6R7, cyano, and C(O)R3;
R3 at each occurrence is independently selected from hydrogen, alkyl, alkenyl, alkynyl, haloalkyl, arylalkyl, aryl, heteroaryl, heterocycle, —OR6, and —NR8R9;
R5 is independently selected from hydrogen, heteroalkyl, C0-C6alkyl-cyano, alkyl, alkenyl, alkynyl, F, Cl, Br, aryl, arylalkyl, heteroaryl, heteroarylalkyl, heterocycle, heterocycloalkyl, haloalkoxy, —O-alkenyl, —O-alkynyl, C0-C6alkyl-OR6, C0-C6alkyl-SR6, C0-C6alkyl-NR6R7, C0-C6alkyl-C(O)R3, C0-C6alkyl-S(O)R3, C0-C6alkyl-C(S)R3, C0-C6alkyl-S(O)2R3, C0-C6alkyl-N(R8)—C(O)R3, C0-C6alkyl-N(R8)—S(O)R3, C0-C6alkyl-N(R8)—S(O)2R3, C0-C6alkyl-O—C(O)R3, C0-C6alkyl-O—S(O)R3, C0-C6alkylN3, and C0-C6alkyl-O—S(O)2R3, each of which is optionally substituted with 1, 2, 3, or 4 substituents independently selected from C1-C4alkyl, C1-C4haloalkyl, —OR6, F, Cl, Br, —NR6R7, cyano, nitro, and C(O)R3, wherein the optional substituent is selected such that a stable compound results;
R6 and R7 are independently selected at each occurrence from hydrogen, alkyl, arylalkyl, heteroarylalkyl, aryl, haloalkyl, heteroaryl, heterocycle, -alkyl-OR8, -alkyl-NR8R9, C(O)R3, S(O)R3, C(S)R3, and S(O)2R3; and
R8 and R9 are independently selected at each occurrence from hydrogen, heteroalkyl, alkyl, arylalkyl, heteroarylalkyl, alkenyl, alkynyl, aryl, heteroaryl, and heterocycle.
|