| CPC A61K 35/766 (2013.01) [A61K 31/225 (2013.01); A61P 35/00 (2018.01); C07C 69/60 (2013.01); C12N 7/00 (2013.01); A61K 2039/585 (2013.01); C12N 2710/10332 (2013.01); C12N 2710/10351 (2013.01); C12N 2740/16134 (2013.01); C12N 2760/16134 (2013.01); C12N 2760/16151 (2013.01); C12N 2760/20232 (2013.01); C12N 2760/20251 (2013.01)] | 18 Claims |
|
1. A method of enhancing production, infection, growth, spread, or titer of an interferon-sensitive virus in an immortalized cell, a cancer cell or a tumor cell, the method comprising:
administering a compound of Formula (I), or a pharmaceutically acceptable salt or solvate thereof,
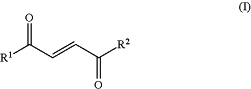 wherein R1 and R2 are each independently OH, (C1-2)alkoxy or linear, branched or cyclic, saturated or unsaturated (C3-20)alkoxy; said alkoxy is optionally substituted with one or more halogen, OH, (C1-4) alkyl, nitro or cyano;
wherein when one of R1 and R2 is OH, the other of R1 and R2 is not OH,
to the immortalized, cancer or tumor cell before, concurrently with, or after infection of the immortalized, cancer or tumor cell with the interferon-sensitive virus.
|