| CPC C07K 14/575 (2013.01) [A61K 9/0019 (2013.01); A61K 38/17 (2013.01); A61K 38/1709 (2013.01); A61K 38/22 (2013.01); A61K 38/26 (2013.01); A61K 45/06 (2013.01); A61K 47/60 (2017.08); A61K 47/64 (2017.08); A61K 47/68 (2017.08); A61K 47/6811 (2017.08); A61K 47/6845 (2017.08); A61K 47/6883 (2017.08); A61K 49/0032 (2013.01); A61K 49/0052 (2013.01); A61K 49/0056 (2013.01); A61P 3/00 (2018.01); A61P 3/04 (2018.01); A61P 3/06 (2018.01); A61P 3/08 (2018.01); A61P 3/10 (2018.01); C07K 1/061 (2013.01); C07K 1/1072 (2013.01); C07K 1/12 (2013.01); C07K 1/18 (2013.01); C07K 5/0205 (2013.01); C07K 14/57545 (2013.01); C07K 16/00 (2013.01); C07K 16/24 (2013.01); A61K 38/00 (2013.01); A61K 2039/505 (2013.01); C07K 2317/14 (2013.01); C07K 2317/21 (2013.01); C07K 2317/24 (2013.01); C07K 2317/33 (2013.01); C07K 2317/52 (2013.01); C07K 2317/55 (2013.01); C07K 2317/56 (2013.01); C07K 2317/565 (2013.01); C07K 2317/567 (2013.01); C07K 2317/71 (2013.01); C07K 2317/92 (2013.01); C07K 2317/94 (2013.01); C07K 2319/30 (2013.01)] | 15 Claims |
|
1. A pharmaceutical composition comprising:
a. a compound of Formula I:
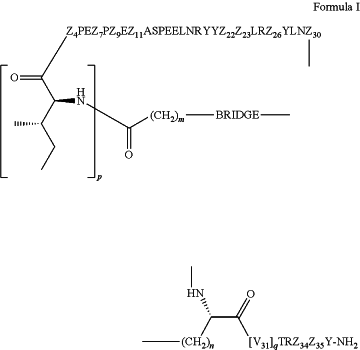 wherein
p is 0 or 1;
m is 0, 1, 2, 3, 4, or 5;
n is 1, 2, 3, or 4;
q is 0 or 1; provided that q is 1 only when Z30 is absent;
BRIDGE is -Ph-CH2—S—, -triazolyl-, —NHC(O)CH2S—, —SCH2C(O)NH—, —(OCH2CH2)2NHC(O)CH2S, —NHC(O)—, or —CH2S—;
Z4 is K, A, E, S, or R;
Z7 is A or K;
Z9 is G or K;
Z11 is D or K;
Z22 is A or K;
Z23 is S or K;
Z26 is A or H;
Z30 is L, W, absent, or K;
provided that Z30 is absent only when q is 1;
Z34 is
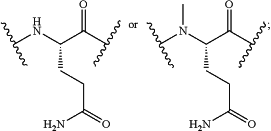 Z35 is
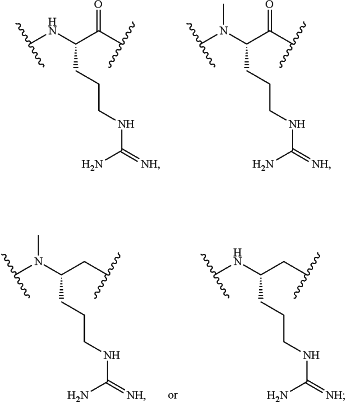 or a derivative thereof; wherein the derivative is the compound of Formula I that is modified by one or more processes comprising amidation, glycosylation, carbamylation, sulfation, phosphorylation, cyclization, lipidation, or pegylation; or a pharmaceutically acceptable salt thereof; and
b. an additional compound selected from the group consisting of a biguanide, insulin, oxyntomodulin, a sulfonylurea, and a thiazolidinedione.
|