| CPC A61B 7/04 (2013.01) [A61B 5/0205 (2013.01); A61B 5/686 (2013.01); A61B 5/725 (2013.01); A61B 5/7282 (2013.01); A61B 7/023 (2013.01); A61N 1/36514 (2013.01); A61N 1/36585 (2013.01); A61B 5/0809 (2013.01); A61B 5/0816 (2013.01); A61B 5/113 (2013.01); A61B 2562/0219 (2013.01); A61B 2562/0247 (2013.01); A61N 1/025 (2013.01); A61N 1/37211 (2013.01); A61N 1/39622 (2017.08)] | 18 Claims |
|
1. An implantable medical device (IMD), comprising:
one or more sensors configured to be implanted in a subject and to sense heart sound signals of the subject;
a respiration sensor configured to sense respiration signals of the subject;
a memory configured to store program instructions; and
a processor that, when executing the program instructions, is configured to:
identify S2 signal segments from the heart sound signals;
analyze the S2 signal segments to identify corresponding pulmonary valve signals (P2 signals) and aortic valve signals (A2 signals);
identify expiration and inspiration phases from the respiration signals;
determine an expiration time interval between the corresponding A2 and P2 signals during the expiration phase;
determine an inspiration time interval between the corresponding A2 and P2 signals during the inspiration phase;
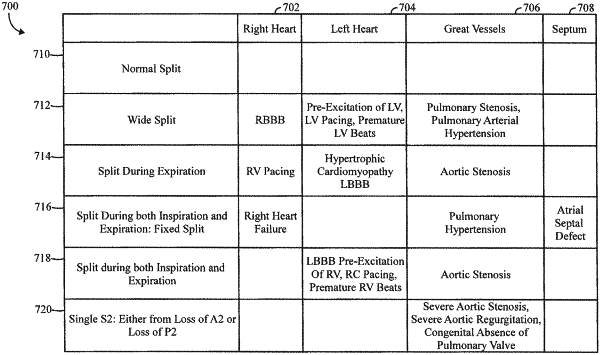
characterize the S2 signal segments to exhibit a first type of S2 split based on a relation between the expiration and inspiration time intervals; and
identify a cardiac condition based on the first type of the S2 split, where the cardiac condition is utilized to determine at least one of a therapy or change in therapy for the subject.
|