| CPC C08B 37/0084 (2013.01) [A01N 65/03 (2013.01); A61K 31/734 (2013.01); A61P 11/12 (2018.01); A61P 31/04 (2018.01)] | 21 Claims |
|
1. A method of:
(a) reducing microbial load on a surface comprising applying a compound comprising a nitric oxide donor to a surface contaminated with a plurality of microbes;
wherein the nitric oxide donor generates nitric oxide and induces oxidative or nitrosative damage to microbial DNA and membrane structures, thereby reducing microbial load, and
wherein said plurality of microbes comprises two or more of the following: gram-positive bacteria, gram-negative bacteria, fungi, yeast, and viruses;
(b) treating a microbial infection comprising, contacting a surface contaminated with a plurality of microbes with a compound comprising a nitric oxide donor;
wherein the nitric oxide donor generates nitric oxide and induces damage to the membrane or DNA of the microbes, thereby reducing the number of viable microbes and treating the infection, and
wherein said plurality of microbes comprises one or more of viruses, gram positive bacteria, gram negative bacteria, drug resistant bacteria, molds, yeasts, fungi, and combinations thereof;
(c) reducing the viscoelasticity of a mucus layer, comprising contacting a surface comprising a mucus layer with a compound comprising a nitric oxide donor;
wherein the nitric oxide donor generates nitric oxide and
wherein the nitric oxide disrupts mucin-mucin interactions in the mucus layer, thereby decreasing mucus viscoelasticity;
or,
(d) delivering nitric oxide to a target site at an anti-microbial concentration for a period of at least 1 to 15 hours, comprising contacting a target site contaminated with a microbial load with a compound comprising a nitric oxide donor;
wherein the nitric oxide donor generates nitric oxide at a concentration sufficient to reduce the number of viable microbes at the target site;
wherein,
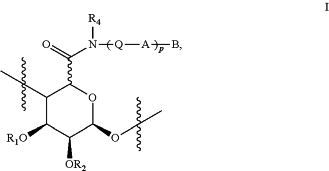
the compound in each of (a), (b), (c), and (d) is a functionalized alginate comprising one or more covalently modified monomers of Formula I
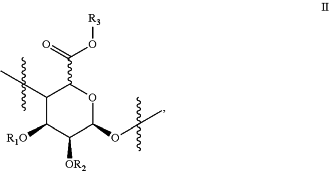
 and optionally, one or more monomers of Formula II
 wherein,
R1 and R2 are each independently selected from the group consisting of hydrogen, C1-5 alkyl(C═O)—, and C1-5 alkyl;
R3 is hydrogen or C1-5 alkyl;
R4 is, in each instance, hydrogen or C1-5 alkyl;
Q is —(CRaRb)v—;
wherein Ra and Rb are independently hydrogen or C1-5 alkyl; and v is an integer from 2 to 6;
A is
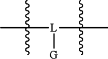 wherein, L is S, O, or N; and
G, in each instance, is hydrogen, is taken together with L to form a nitric oxide donor, or is absent;
wherein the nitric oxide donor is selected from the group consisting of a diazeniumdiolate, a nitrosamine, a hydroxyl amine, a hydroxyurea, a nitrosothiol, a hydroxyl nitrosamine, and a combination thereof;
p is an integer from 1 to 10;
B is selected from the group consisting of hydrogen, —Y—Z, and C1-5 alkyl, wherein said C1-5 alkyl is optionally substituted with amino, hydroxyl, nitrile, CO2H, mono(C1-6)alkylamino-, di(C1-6)alkylamino-, —(CO)NRcRd, or —NRc(CO)Rd,
wherein Rc and Rd are each independently selected from the group consisting of hydrogen and C1-6 alkyl,
wherein Y has a structure of:
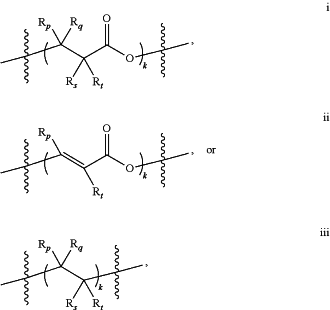 wherein Rp, Rq, Rs and Rt, in each instance, are independently, hydrogen or hydroxyl; and
k is an integer from 1 to 20; and
Z has a structure of:
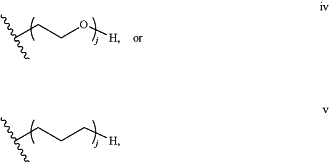 wherein j, in each instance, is an integer from 1 to 100; and
wherein the functionalized alginate comprises at least one monomer of Formula I containing the nitric oxide donor.
|