| CPC G01N 27/308 (2013.01) [B01L 3/502715 (2013.01); B29C 64/112 (2017.08); B29C 64/30 (2017.08); B33Y 10/00 (2014.12); B33Y 40/20 (2020.01); B33Y 80/00 (2014.12); B41M 1/12 (2013.01); B41M 1/30 (2013.01); B41M 3/006 (2013.01); G01N 27/333 (2013.01); G01N 27/48 (2013.01); B01L 2300/0645 (2013.01); B01L 2300/0816 (2013.01); B01L 2300/16 (2013.01); B29L 2031/752 (2013.01)] | 9 Claims |
|
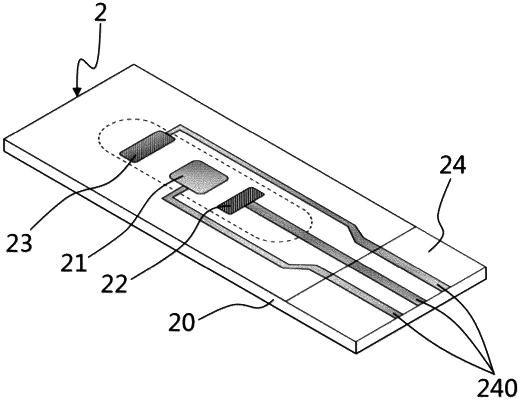
1. An electrode-modified heavy metal ion microfluidic detection chip, including a microfluidic module (1) and a three-electrode sensor (2), wherein:
the microfluidic module (1) comprises a micro-channel (10), an inlet duct (12) and an outlet duct (13) are provided at opposite ends of the at least one micro-channel (10); the three-electrode sensor (2) comprises three all-solid-state planar electrodes provided on a card-shaped base plate (20), a working electrode (21), an auxiliary electrode (22) and a reference electrode (23); one end of the three-electrode sensor (2) is an interface area (24), contacts (240) at terminals of leads of the three all-solid-state planar electrodes are arranged in the interface area; a sensor slot (11) matching the three-electrode sensor (2) is provided at a bottom portion of the micro-channel (10) in the microfluidic module (1); when the three-electrode sensor (2) is inserted in the sensor slot (11), the three all-solid-state planar electrodes are communicated with the micro-channel (10), the three-electrode sensor (2) is assembled with the microfluidic module (1) by being inserted into the sensor slot (11), and is detachable; the interface area (24) is outside the sensor slot (11);
the microfluidic module (1) is a 3D printed transparent flexible piece, the micro-channel (10), the inlet duct (12), the outlet duct (13), and the sensor slot (11) are integrally printed and formed on with the microfluidic module (1); the working electrode (21) is a bare carbon electrode, and a surface thereof is modified by porous nano-NiMn2O4; the auxiliary electrode (22) is an Ag electrode; and the reference electrode (23) is an Ag/AgCl electrode;
wherein preparing the porous nano-NiMn2O4, specific steps of preparing the porous nano-NiMn2O4 include:
dissolving MnCl2·6H2O 20 mmol/L, NiCl2·6H2O 40 mmol/L, Mn(NH2)2 120 mmol/L, and NH4F 0.1 g in ethanol 5 mL and deionized water 30 mL, stirring violently for 30 min;
cooling the solution to room temperature, cleaning reaction products with distilled water for at least 5 times, and drying the same; and
giving air annealing treatment to the reaction products at a tubular furnace at 2° C./min, preserving for 3 h at 350° C., so as to obtain porous nano-NiMn2O4 powder.
|