| CPC A61M 16/0003 (2014.02) [A61M 16/0051 (2013.01); A61M 16/0066 (2013.01); A61M 16/026 (2017.08); A61M 16/0672 (2014.02); A61M 16/0816 (2013.01); A61M 16/105 (2013.01); A61M 16/125 (2014.02); A61M 16/161 (2014.02); A61M 16/201 (2014.02); A61M 2016/003 (2013.01); A61M 2016/1025 (2013.01); A61M 2205/3375 (2013.01); A61M 2205/583 (2013.01); A61M 2230/005 (2013.01); A61M 2230/06 (2013.01); A61M 2230/205 (2013.01); A61M 2230/42 (2013.01); A61M 2230/435 (2013.01)] | 17 Claims |
|
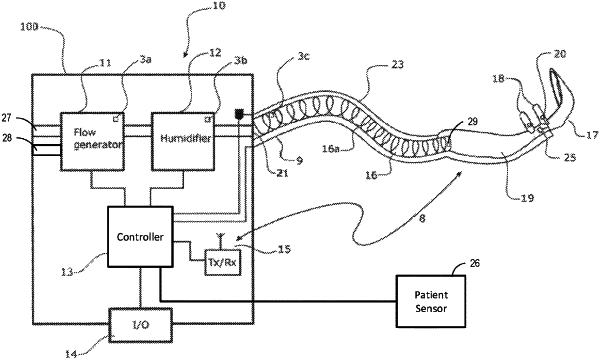
1. A respiratory apparatus that provides a flow of gases to a patient, wherein the respiratory apparatus is configured to deliver high flow therapy to the patient, the respiratory apparatus comprising:
a gases composition sensor configured to determine at least a measured fraction of delivered oxygen content (FdO2) of gases flow during operation of the respiratory apparatus, wherein the gases composition sensor is an ultrasonic sensor system;
a controller configured to control delivery of gases to the patient using closed loop control, wherein the controller is configured to:
receive patient parameter data indicative of oxygen saturation (SpO2) of the patient from at least one sensor;
execute a control phase, wherein operation of the respiratory apparatus during a therapy session is based at least in part on the patient parameter data, wherein the control phase includes determining a target FdO2, wherein the target FdO2 is determined based at least in part on a target SpO2, measured SpO2, measured FdO2, and a previous target FdO2.
|