| CPC C07D 417/14 (2013.01) [A61P 35/00 (2018.01)] | 30 Claims |
|
1. A compound of Formula (I) or a pharmaceutically acceptable salt thereof;
wherein:
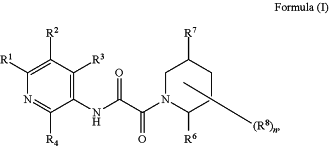 each R1 is independently selected from H, -D, halo, —CN, —C1-C6 alkyl, —C1-C6 heteroalkyl, —C1-C6 haloalkyl, —C3-C9 cycloalkyl, 3-10 membered heterocyclyl, heterocyclylalkyl, heteroarylalkyl, arylalkyl, cycloalkylalkyl, —N(Ra1)2, —C(═O)Ra1, —C(═O)ORa1, —NRa1C(═O)Ra1, —NRa1C(═O)ORa1, —C(═O)N(Ra1)2, —OC(═O)N(Ra1)2, —S(═O)Ra1, —S(═O)2Ra1, —SRa1, —S(═O)(═NRa1)Ra1, —NRa1S(═O)2Ra1 and —S(═O)2N(Ra1)2;
each R2 is independently selected from halo, —CN, —C1-C6 alkyl, —C1-C6 heteroalkyl, —C1-C6 haloalkyl, —C1-C6 haloalkoxy, —C3-C9 cycloalkyl, 3-10 membered heterocyclyl, heterocyclylalkyl, heteroarylalkyl, arylalkyl, cycloalkylalkyl, —ORa2, —N(Ra2)2, —C(═O)Ra2, —C(═O)ORa2, —NRa2C(═O)Ra2, —NRa2C(═O)ORa2, —C(═O)N(Ra2)2, —C(═O)N(ORa2)(Ra2), —OC(═O)N(Ra2)2, —S(═O)Ra2, —S (═O)2Ra2, —SRa2, —S(═O)(═NRa2)Ra2, NRa2S(═O)2Ra2, —S(═O)2N(Ra2)2;
each R3 is independently selected from H, -D, halo, —CN, —C1-C6 alkyl, —C1-C6 heteroalkyl, —C1-C6 haloalkyl, —C3-C9 cycloalkyl, 3-10 membered heterocyclyl, heterocyclylalkyl, heteroarylalkyl, arylalkyl, cycloalkylalkyl, —ORa3, —N(Ra3)2, —C(═O)Ra3, —C(═O)ORa3, —NRa3C(═O)Ra3, —NRa3C(═O)ORa3, —C(═O)N(Ra3)2, —OC(═O)N(Ra3)2, —S(═O)Ra3, —S(═O)2Ra3, —SRa3, —S(═O)(═NRa3)Ra3, —NRa3S(═O)2Ra3 and —S(═O)2N(Ra3)2;
each R4 is independently selected from H, -D, halo, —CN, —C1-C6 alkyl, —C1-C6 heteroalkyl, —C1-C6 haloalkyl, —C3-C9 cycloalkyl, 3-10 membered heterocyclyl, heterocyclylalkyl, heteroarylalkyl, arylalkyl, cycloalkylalkyl, —ORa4, —N(Ra4)2, —C(═O)Ra4, —C(═O)ORa4, —NRa4C(═O)Ra4, —NRa4C(═O)ORa4, —C(═O)N(Ra4)2, —OC(═O)N(Ra4)2, —S(═O)Ra4, —S(═O)2Ra4, —SRa4, —S(═O)(═NRa4)Ra4, —NRa4 and —S(═O)2N(Ra4)2;
each R6 is independently absent or selected from H, -D, halo, —CN, —C1-C6 alkyl, —C1-C6 heteroalkyl, —C1-C6 haloalkyl, —C3-C10 carbocyclyl, 3-10 membered heterocyclyl, heterocyclylalkyl, C6-C10 aryl, 5-10 member heteroaryl, heteroarylalkyl, arylalkyl, cycloalkylalkyl, —ORa6, —N(Ra6)2, —C(═O)Ra6, —C(═O)ORa6, —NRa6C(═O)Ra6, NRa6C(═O)ORa6, —C(═O)N(Ra6)2, —OC(═O)N(Ra6)2, —S(═O)Ra6, —S(═O)2Ra6, —SRa6, —S(═O)(═NRa6)Ra6, NRa6S(═O)2Ra6 and —S(═O)2N(Ra6)2, wherein each alkyl, carbocyclyl, heterocyclyl, cycloalkylalkyl, heterocyclylalkyl, aryl, heteroaryl, arylalkyl and heteroarylalkyl is optionally substituted at any available position;
each R7 is independently absent or selected from H, -D, halo, —CN, —C1-C6 alkyl, —C1-C6 hydroxyalkyl, haloalkyl, —C3-C9 cycloalkyl, 3-10 membered heterocyclyl, 5-6-membered monocyclic heteroaryl, heterocyclylalkyl, heteroarylalkyl, arylalkyl, cycloalkylalkyl, —ORa7, —N(Ra7)2, —C(═O)Ra7, —C(═O)ORa7, —NRa7C(═O)Ra7, —NRa7C(═O)ORa7, —C(═O)N(Ra7)2, —OC(═O)N(Ra7)2, —S(═O)Ra7, —S(═O)2Ra7, —SRa7, —S(═O)(═NRa7)Ra7, —NRa7S(═O)2Ra7 and —S(═O)2N(Ra7)2;
each R8 is independently selected from H, -D, ═O, halo, —CN, —C1-C6 alkyl, —C1-C6 heteroalkyl, —C1-C6 haloalkyl, —C3-C9 cycloalkyl, 3-10 membered heterocyclyl, heterocyclylalkyl, heteroarylalkyl, arylalkyl, cycloalkylalkyl, —ORa8, —N(Ra8)2, —C(═O)Ra8, C(═O)ORa8, —NRa8C(═O)Ra8, —ORa8, —CH2C(═O)N(Ra8)2, —C(═O)N(Ra8)2, —OC(═O)N(Ra8)2, —CH2C(═O)N(Ra8)2, —S(═O)Ra8, —S(═O)2Ra8, —SRa8, —S(═O)(═NRa8)Ra8, —NRa8S(═O)2Ra and —S(═O)2N(Ra8)2;
each Ra1, Ra2, Ra3, Ra4, Ra6, Ra7 and Ra8 is independently selected from H, C1-C6 alkyl, —C1-C6 heteroalkyl, C3-C9 cycloalkyl, 3-7 membered heterocyclyl, cycloalkylalkyl, heterocyclylalkyl, aryl, 5-6 membered heteroaryl, arylalkyl and heteroarylalkyl wherein each alkyl, cycloalkyl, heterocyclyl, cycloalkylalkyl, heterocyclylalkyl, aryl, heteroaryl, arylalkyl and heteroarylalkyl is optionally substituted at any available position; and
n is 0, 1, 2 or 3 provided that:
(i) when R1 is H, R2 is not halo, —OPr, —N(CH3)2 or —CF3;
(ii) when R1 is ORa1, R2 is not —ORa2;
(iii) when R1 is H and R2 is —CH3, R8 groups cannot be taken together to form a ring and R6 is not absent or H, and is not thiazolyl, furanyl or pyrrolyl;
(iv) when R2 is Me, R1 is not piperidine
(v) the compound is not:
A. 5-(2-(5-methyl-2-(p-tolyl)piperidin-1-yl)-2-oxoacetamido)nicotinamide or any of its enantiomers or diastereomers;
B. 2-(2-(4-(2H-tetrazol-5-yl)phenyl)-5-methylpiperidin-1-yl)-N-(5,6-dimethylpyridin-3-yl)-2-oxoacetamide or any of its enantiomers or diastereomers;
C. 2-cyano-5-(2-(5-methyl-2-phenylpiperidin-1-yl)-2-oxoacetamido)nicotinamide or any of its enantiomers or diastereomers.
|