| CPC G16H 40/63 (2018.01) [G06K 7/10415 (2013.01); G06Q 10/087 (2013.01); G06Q 10/0875 (2013.01); G06Q 10/10 (2013.01); G16H 15/00 (2018.01); G16H 20/10 (2018.01); G16H 40/40 (2018.01); G16H 40/67 (2018.01); G16Z 99/00 (2019.02); H04W 4/80 (2018.02)] | 37 Claims |
|
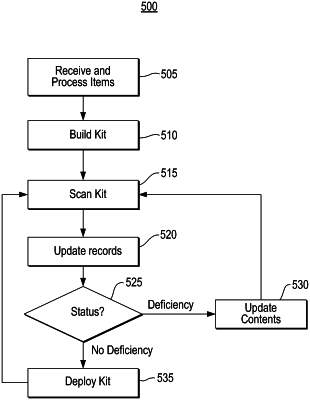
1. A system for managing pharmacy kits, comprising:
a reading station, comprising:
a pharmacy kit container formed of a material designed to provide electromagnetic shielding,
wherein each of the pharmacy kits is a transportable container having a collection of pharmacy items for a common purpose that can be deployed for a specific medical procedure, for a specific physician, or to a designated location, and
wherein the pharmacy kit container is configured to temporarily enclose the pharmacy kit for verification, and
an antenna coupled to the pharmacy kit container;
computer-executable instructions that when executed by a processor cause the processor to:
cause the antenna to emit a radio signal at least within the pharmacy kit container, and
receive tag information from a plurality of radio frequency identification (RFID) tags coupled to a plurality of medicinal containers configured to store a plurality of drugs, the plurality of RFID tags being associated with a pharmacy kit located within the pharmacy kit container,
wherein for the plurality of RFID tags, a particular RFID tag is coupled to a particular medicinal container of the plurality of medicinal containers and the particular medicinal container is configured to store a particular drug of the plurality of drugs, and
wherein the particular RFID tag is associated with drug data of the particular drug, the drug data comprising at least an identifier of the particular drug and an expiration of the particular drug; and
an information processing system communicatively coupled to the reading station and configured to:
verify a first drug of the plurality of drugs using the tag information, wherein to verify the first drug, the information processing system is configured to:
determine whether a first medicinal container associated with the first drug is present based at least in part on the tag information received from the plurality of RFID tags,
based at least upon a determination that the first medicinal container is not present in the pharmacy kit, determine whether a second medicinal container configured to store a substitute first drug is present based at least in part on the tag information,
based at least upon a determination that the second medicinal container is present, determine whether an expiration of the substitute first drug satisfies an expiration threshold based at least in part on the drug data associated with a first RFID tag of the plurality of RFID tags, wherein the first RFID tag is coupled to the second medicinal container, and
based at least upon a determination that the expiration of the substitute first drug satisfies the expiration threshold, verify the first drug,
identify any drugs that are missing from the pharmacy kit based at least in part on the tag information received from the plurality of RFID tags and a template for the pharmacy kit,
identify any drugs of the plurality of drugs that do not satisfy the expiration threshold based at least in part on the tag information received from the plurality of RFID tags, and
cause a display to display information regarding the plurality of drugs in the pharmacy kit including the substitute first drug, the any drugs that are missing from the pharmacy kit, and the any drugs of the plurality of drugs that do not satisfy the expiration threshold.
|