| CPC A61B 17/0057 (2013.01) [A61B 17/00491 (2013.01); A61B 17/06166 (2013.01); A61B 17/3401 (2013.01); A61L 31/129 (2013.01); A61M 5/3293 (2013.01); A61B 2017/00004 (2013.01); A61B 2017/00659 (2013.01); A61B 2017/06176 (2013.01)] | 15 Claims |
|
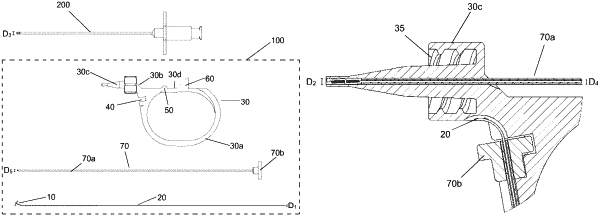
1. A dural sealing system (100) comprising:
an implant (10) joined to a guiding thread (20) of diameter D1;
an introductory device (70) comprising a tube (70a) having an outside diameter D5, the tube (70a) having:
(i) an interior hollow section of diameter D4, wherein D4 is greater than D1,
(ii) a first free end, and
(iii) a second end having a stop (70b) positioned on said second end; and
a transfer device (30) comprising a grip portion (30a) and a nozzle (30c), wherein the nozzle comprises a first nozzle end (30c) and a second nozzle end (30d) with a hollow section (30b), of diameter D2, connecting the first and second nozzle ends, wherein the first nozzle end (30c) comprises an epidural needle coupling and the second nozzle end (30d) accommodates insertion of the tube (70a) of the introductory device (70) into the hollow section (30b), wherein the diameter of the hollow section (30b) is greater than the outside diameter D5 of the tube (70a), and wherein the grip portion comprises a notch (40) that accommodates the stop (70b) of the introductory device (70) when the tube (70a) is positioned in the hollow section (30b) of the nozzle (30c) and the first free end of the tube (70a) is positioned proximate to the first nozzle end (30c),
wherein the guiding thread (20) is co-axially positioned inside the interior hollow section of the tube (70a).
|