| CPC A61M 5/1723 (2013.01) [G06Q 30/04 (2013.01); G16H 10/20 (2018.01); G16H 20/17 (2018.01); G16H 40/20 (2018.01); G16H 40/40 (2018.01); G16H 40/67 (2018.01); G16H 50/20 (2018.01); G16H 50/30 (2018.01); G16H 50/50 (2018.01); G16H 50/70 (2018.01); A61M 2205/3553 (2013.01); A61M 2205/502 (2013.01); A61M 2205/52 (2013.01); A61M 2205/581 (2013.01); A61M 2205/582 (2013.01); A61M 2205/8206 (2013.01); A61M 2230/201 (2013.01)] | 20 Claims |
|
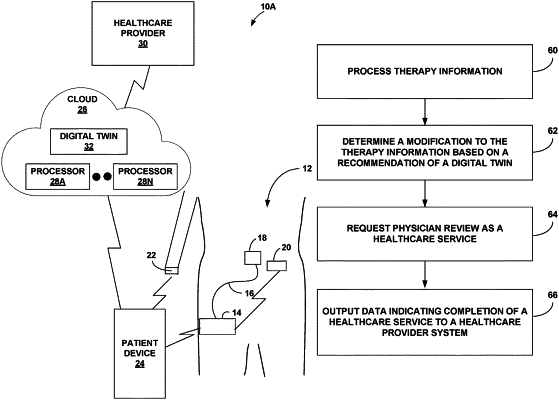
1. A system for therapy delivery, the system comprising:
a delivery device configured to execute therapy information for directing the therapy delivery for a patient, wherein the therapy information comprises a treatment schedule specifying one or more treatments over a time period, wherein for each of the one or more treatments, the therapy information comprises a treatment type and a treatment amount; and
processing circuitry communicably coupled to the delivery device, the processing circuitry configured to:
determine a modification to the therapy information based on a recommendation of a digital twin, wherein the digital twin determines an estimated glucose level expected to be within a healthy range if the delivery device executes the modification to therapy information; and
in response to physician review of the modification to the therapy information, output, to a healthcare provider system for the patient, data indicating completion of a healthcare service corresponding to the physician review.
|