| CPC A61F 2/2472 (2013.01) [A61F 2/82 (2013.01); A61M 60/117 (2021.01); G01N 15/0606 (2013.01); G01N 2015/018 (2024.01); G01N 15/075 (2024.01); G01N 33/4905 (2013.01)] | 5 Claims |
|
1. A method for an in vitro simulation and an evaluation of platelet adhesion in a blood-contacting medical device, comprising the following steps:
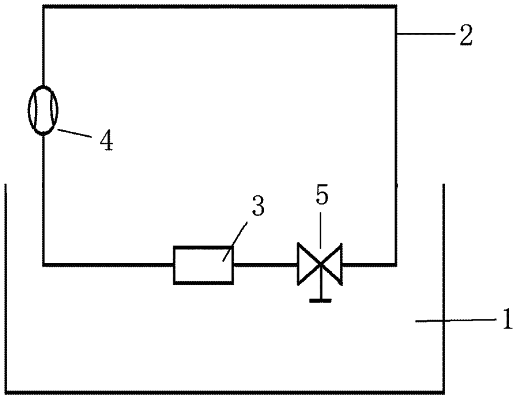
(1) using a glycerin aqueous solution with a mass percentage concentration of 40% in an extracorporeal circulation circuit to simulate a viscosity and hydrodynamic characteristics of blood, and adding fluorescent particles with a diameter of 3 μm to 5 μm to the glycerin aqueous solution to simulate platelets, to obtain a simulated blood solution;
(2) after the simulated blood solution circulates in the extracorporeal circulation circuit for 24 h, removing flow passage components of the blood-contacting medical device, and observing a deposition of the fluorescent particles on a blood-contacting surface inside the blood-contacting medical device by naked eyes and photographs; and
(3) using a laser-induced fluorescence (LIF) technique to apply laser light on the blood-contacting surface to obtain laser-induced fluorescent particles, wherein the blood-contacting surface is deposited with the fluorescent particles, and using a charge-coupled device (CCD) camera imaging method to photograph an aggregation and adhesion of the laser-induced fluorescent particles.
|