| CPC G01N 33/6896 (2013.01) [G01N 2800/2821 (2013.01)] | 35 Claims |
|
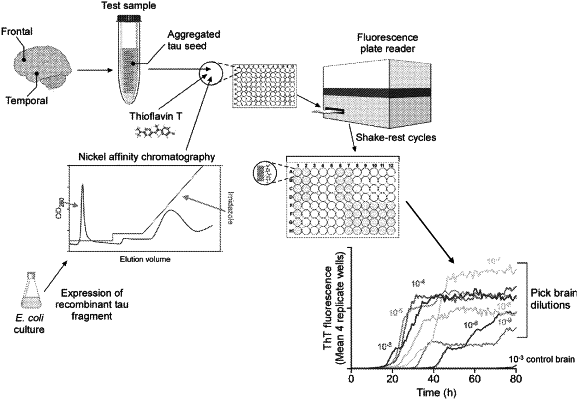
1. A method of determining whether a subject has a 3R/4R Tauopathy, comprising:
(a) performing a seeded Tau polymerization assay on a biological sample from the subject, comprising:
(i) contacting the biological sample with a purified recombinant truncated Tau protein, wherein the recombinant truncated Tau protein consists of SEQ ID NO: 43 or SEQ ID NO: 44, to form a reaction mixture;
(ii) incubating the reaction mixture to permit coaggregation of disease-associated Tau protein (TD) present in the biological sample with the recombinant truncated Tau protein;
(iii) maintaining incubation conditions that promote coaggregation of the recombinant truncated Tau protein with the TD to result in a conversion of the recombinant truncated Tau protein to amyloid Tau protein aggregates while inhibiting development of spontaneously forming Tau protein aggregates (rT(spon)); and
(iv) agitating amyloid Tau protein aggregates formed during step (iii), wherein the agitating comprises shaking the reaction mixture in a shaking cycle, wherein each shaking cycle comprises a period of rest and a period of shaking; and
(b) detecting amyloid Tau protein present in the reaction mixture, and wherein detection of amyloid Tau protein in the reaction mixture indicates that the subject has the 3R/4R Tauopathy.
|