| CPC C07D 403/14 (2013.01) [A61P 37/06 (2018.01)] | 29 Claims |
|
1. A method of treating viral diseases in a patient in need thereof, comprising administering to said patient a therapeutically effective amount of a compound of Formula I:
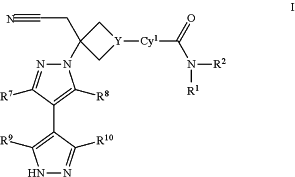 or a pharmaceutically acceptable salt thereof; wherein:
Cy1 is phenyl, pyridyl, pyrimidinyl, pyrazinyl, or pyridazinyl, each of which is optionally substituted by 1, 2, 3, or 4 groups independently selected from R3, R4, R5, and R6;
Y is N or CH;
R1 is C1-6 alkyl, C1-6 haloalkyl, C3-7 cycloalkyl, C3-7 cycloalkyl-C1-3 alkyl, 4-7 membered heterocycloalkyl, 4-7 membered heterocycloalkyl-C1-3 alkyl, phenyl, phenyl-C1-3 alkyl, 5-6 membered heteroaryl or 5-6 membered heteroaryl-C1-3 alkyl, each of which is optionally substituted with 1, 2, or 3 substituents independently selected from fluoro, chloro, C1-3 alkyl, —OH, —O(C1-3 alkyl), —CN, —CF3, —CHF2, —CH2F, —NH2, —NH(C1-3 alkyl), —N(C1-3 alkyl)2, —C(═O)N(C1-3 alkyl)2, —C(═O)NH(C1-3 alkyl), —C(═O)NH2, —C(═O)O(C1-3 alkyl), —S(═O)2(C1-3 alkyl), —S(═O)2(C3-6 cycloalkyl), —C(═O)(C3-6 cycloalkyl), and —C(═O)(C1-3 alkyl);
R2 is H or C1-3 alkyl; wherein said C1-3 alkyl is optionally substituted by 1, 2, or 3 substituents independently selected from fluoro, chloro, —OH, —O(C1-3 alkyl), —CN, —CF3, —CHF2, —CH2F, NH2, —NH(C1-3 alkyl), and —N(C1-3 alkyl)2; or
R1 and R2, together with the nitrogen atom to which they are attached, form a 4-, 5- or 6-membered heterocycloalkyl ring, which is optionally substituted with 1, 2, or 3 substitutents independently selected from F, Cl, —OH, —O(C1-3 alkyl), —CN, C1-3 alkyl, C1-3 haloalkyl, —NH2, —NH(C1-3 alkyl), —N(C1-3 alkyl)2, —CH2CN, and —CH2OH;
R3 is H, F, Cl, —CN, C1-3 alkyl, C1-3 fluoroalkyl, —O(C1-3 alkyl), or —O(C1-3 fluoroalkyl);
R4 is H, F, Cl, —CN, C1-3 alkyl, C1-3 fluoroalkyl, —O(C1-3 alkyl), or —OC(C1-3 fluoroalkyl);
R5 is H, F, Cl, —CN, C1-3 alkyl, C1-3 fluoroalkyl, —O(C1-3 alkyl), or —OC(C1-3 fluoroalkyl);
R6 is H, F, Cl, —CN, C1-3 alkyl, C1-3 fluoroalkyl, —O(C1-3 alkyl), or —OC(C1-3 fluoroalkyl);
R7 is H, F, Cl, C1-3 alkyl, C1-3 haloalkyl, —
NR17R17a, —NHC(═O)R17b,—C(═O)NR17aR17b, —NHS(═O)2R17b, or —S(═O)2NR17aR17b, wherein said C1-3 alkyl is optionally substituted with 1, 2, or 3 substituents selected from F, Cl, —CN, —CF3, —CHF2, —CH2F, —NH2, —NH(CH3), —N(CH3)2, OH, —OCH3, and —OCF3, —OCHF2, and —OCH2F;
R8 is H, F, Cl, C1-3 alkyl, or C1-3 haloalkyl;
R9 is H, F, Cl, C1-3 alkyl, C1-3 haloalkyl, cyclopropyl, —CN, —NH2, —NH(C1-3 alkyl), or —N(C1-3 alkyl)2, wherein said C1-3 alkyl is optionally substituted with 1, 2, or 3 substituents selected from F, chloro, —CN, —CF3, —CHF2, —CH2F, —NH2, and OH;
R10 is H, F, Cl, C1-3 alkyl, C1-3 haloalkyl, cyclopropyl, —CN, —NH2, —NH(C1-3 alkyl), or —N(C1-3 alkyl)2, wherein said C1-3 alkyl is optionally substituted with 1, 2, or 3 substituents selected from F, chloro, —CN, —CF3, —CHF2, —CH2F, —NH2, and OH;
R17 is C1-6 alkyl, phenyl or 5-6 membered heteroaryl, each of which is optionally substituted with 1, 2, 3 or 4 independently selected R27 substituents;
R17a is H or alkyl;
R17b is C1-3 alkyl optionally substituted with 1, 2, or 3 substituents selected from F, chloro, —CN, —CF3, —CHF2, —CH2F, —NH2, —NH(CH3), —N(CH3)2, OH, —OCH3, and —OCF3, —OCHF2, and —OCH2F; and
each R27 is independently selected from halo, —OH, NO2, —CN, C1-3 alkyl, C2-3 alkenyl, C2-3 alkynyl, C1-3 haloalkyl, cyano-C1-3 alkyl, HO—C1-3 alkyl, CF3—C1-3 hydroxyalkyl, C1-3 alkoxy-C1-3 alkyl, C3-7 cycloalkyl, C1-3 alkoxy, C1-3 haloalkoxy, H2N—, (C1-3 alkyl)NH—, (C1-3 alkyl)2N—, HS—, C1-3 alkyl-S—, C1-3 alkyl-S(═O)—, C1-3 alkyl-S(═O)2—, carbamyl, C1-3 alkylcarbamyl, di(C1-3 alkyl)carbamyl, carboxy, C1-3 alkyl-C(═O)—, C1-4 alkoxy-C(═O)—, C1-3 alkyl-C(═O)O—, C1-3 alkyl-C(═O)NH—, C1-3 alkyl-S(═O)2NH—, H2N—SO2—, C1-3 alkyl-NH—S(═O)2—, (C1-3 alkyl)2N—S(═O)2—, H2N—S(═O)2NH—, C1-3 alkyl-NHS(═O)2NH—, (C1-3 alkyl)2N—S(═O)2NH—, H2N—C(═O)NH—, C1-3 alkyl-NHC(═O)NH—, and (C1-3 alkyl)2N—C(═O)NH—.
|