| CPC C07C 229/66 (2013.01) [C07B 2200/13 (2013.01)] | 30 Claims |
|
1. A crystalline form of a pharmaceutically acceptable salt of the compound of Formula (I):
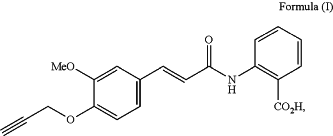 wherein the crystalline form has a water sorption of less than 1.7% at 25° C. and 80% relative humidity as determined by dynamic vapor sorption.
|