| CPC A61M 37/0015 (2013.01) [A61M 2037/0023 (2013.01); A61M 2037/0053 (2013.01); A61M 2037/0061 (2013.01); A61M 2202/0007 (2013.01); A61M 2202/03 (2013.01); A61M 2202/07 (2013.01); A61M 2202/30 (2013.01)] | 20 Claims |
|
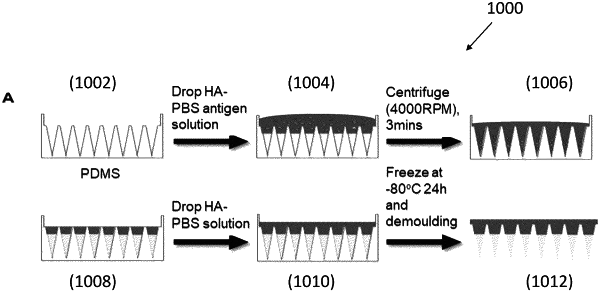
1. A cryo formulation-based microneedle device for transdermal delivery of bioactive therapeutic agents, comprising:
one or more microneedle patches each including an array of miniaturized needles, each miniaturized needle defining a base end and a tip; and
a substrate to which the base end of the array of miniaturized needles is attached or integrated thereto;
wherein the microneedle patches are in a cryo status;
wherein each of the one or more microneedle patches are adapted to be applied on a skin surface, in which the miniaturized needles penetrate into skin;
wherein the miniaturized needles are further arranged to melt so as to release one or more bioactive therapeutic agents into the skin to achieve a targeted therapeutic effect;
wherein each of the one or more microneedle patches consists of a matrix solution and one or more bioactive therapeutic agents; and
wherein the one or more bioactive therapeutic agents includes at least one of a spike glycoprotein, a nucleocapsid protein, mRNA for encoding a spike glycoprotein, mRNA for encoding a nucleocapsid protein, and a combination thereof.
|