| CPC C08G 83/008 (2013.01) [A61L 27/18 (2013.01); A61L 27/56 (2013.01); A61L 27/58 (2013.01); A61L 31/06 (2013.01); A61L 31/146 (2013.01); A61L 31/148 (2013.01); C08G 18/3206 (2013.01); C08G 18/73 (2013.01); C08G 2230/00 (2013.01)] | 19 Claims |
|
1. A process for the manufacture of a supramolecular biomedical polymer for porous biomedical implants, wherein the supramolecular biomedical polymer has an ultimate tensile strength of at least 35 MPa, a Young's modulus of between 40 and 160 MPa, and an elongation at break of at least 350%, as determined by test method ASTM D 1708-96 with a crosshead speed of 20 mm/min, the process comprising reacting a compound F′ according to Formula (1):
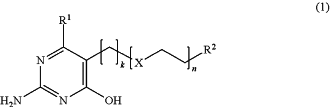 with:
a diisocyanate compound C′ according to the Formula O═C═N—R3—N═C═O;
a functionalized polymer A′ according to the Formula P-(FG1)w; and
a compound B′ according to the Formula FG2-R4-FG3;
wherein:
X is O or S;
k is an integer of 1 to 20;
n is an integer of 0 to 8;
R1 is a C1-C13 alkyl group;
R2 is a functional group selected from OH, SH and NH2;
FG1, FG2 and FG3 are functional groups independently selected from OH and NH2;
w is in the range of about 1.8 to about 2;
functionalized polymer A′ has a number average molecular weight Mn, as determined from its hydroxyl value, of about 1000 to about 2000 Da, and is selected from hydroxy terminated poly(1,6-hexanediol) carbonate, poly(tetramethylene glycol), and FG1-functionalized hydrogenated polybutadiene;
R3 is a linear C4-C20 alkylene group;
R4 is a linear C2-C20 alkylene group; and
wherein the molar ratio of compounds A′, B′, C′ and F′ expressed as A′:B′:C′:F′ is between 1:1.5:3.5:1 and 1:2:4:1.
|