| CPC A61F 7/123 (2013.01) [A61B 5/01 (2013.01); A61B 5/4836 (2013.01); A61B 17/12022 (2013.01); A61B 18/02 (2013.01); A61B 18/0218 (2013.01); A61B 18/04 (2013.01); A61B 5/6853 (2013.01); A61B 2017/00539 (2013.01); A61B 2018/00011 (2013.01); A61B 2018/0022 (2013.01); A61B 2018/00041 (2013.01); A61B 2018/00488 (2013.01); A61B 2018/00553 (2013.01); A61B 2018/00559 (2013.01); A61B 2018/00577 (2013.01); A61B 2018/00791 (2013.01); A61B 2018/00863 (2013.01); A61B 2018/00982 (2013.01); A61B 2018/0212 (2013.01); A61B 2018/0262 (2013.01); A61B 2018/046 (2013.01)] | 20 Claims |
|
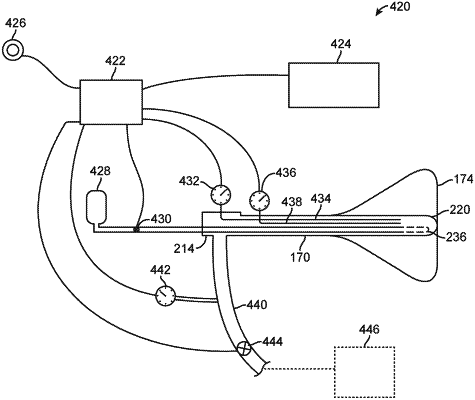
1. A tissue treatment system, comprising:
a handle;
an elongate lumen extending from the handle;
at least one infusion lumen positioned through or along the elongate lumen;
at least one delivery lumen in fluid communication with the infusion lumen, wherein the delivery lumen defines one or more openings for infusing a cryoablative agent;
a liner defining an interior in which the elongate lumen is positionable;
a sheath translatable relative to the elongate lumen;
an exhaust lumen in fluid communication with the interior of the liner; and
a valve positioned within the exhaust lumen, wherein the valve is configured to control a pressure of the cryoablative agent within the system.
|