| CPC G01N 33/5014 (2013.01) [A61B 5/361 (2021.01); A61B 5/363 (2021.01); A61K 31/445 (2013.01); A61K 49/0004 (2013.01); A61P 9/06 (2018.01); G01N 33/6893 (2013.01); A61K 45/06 (2013.01); G01N 2800/326 (2013.01); G01N 2800/50 (2013.01)] | 7 Claims |
|
1. A method for determining whether a medication is associated with an increased risk of drug-induced long QT syndrome or Torsades de Pointes in combination with at least one other drug by determining a drug-specific index for the medication, the method comprising the steps of:
(i) calculating a first index variable by determining;
an IC50 value for block of one or more of IKr and IKs,
a Cmax of the medication at a test dose,
a daily dose amount of the medication,
a protein binding value for the medication at a target protein,
a drug-drug interaction coefficient (DDIC) for the medication, and
calculating a value for the first index variable based on the following equation:
 (ii) calculating a second index variable by determining:
an IC50 value for block of CaV1.2 current,
the IC50 value for block of one or more of IKr and IKs, and
calculating a value for the second index variable based on the following equation:
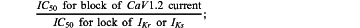 (iii) calculating a third index variable by determining:
an IC50 value for block of NaV1.5 current,
the IC50 value for block of one or more of IKr and IKs, and
calculating a value for the third index variable based on the following equation:
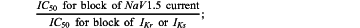 (iv) calculating a value for a fourth index variable based on whether the medication is an inhibitor of hERG trafficking; and
(v) combining the values for the first, second, third, and fourth index variables to provide the drug-specific index, wherein a drug-specific index of less than a predetermined threshold value is indicative of an increased risk of drug-induced long QT syndrome or Torsades de Pointes for the medication in combination with the at least one other drug.
|