| CPC A61K 31/4725 (2013.01) [A61K 31/4985 (2013.01); A61K 31/5025 (2013.01); A61K 31/5377 (2013.01); A61K 31/541 (2013.01); C07D 401/04 (2013.01); C07D 417/14 (2013.01); C07D 471/04 (2013.01); C07D 487/04 (2013.01); C07D 493/08 (2013.01); C07D 513/04 (2013.01)] | 5 Claims |
|
1. A compound having Formula (V)
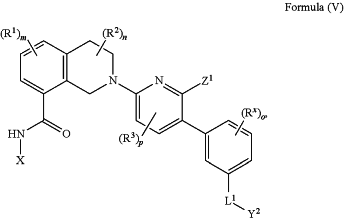 or a therapeutically acceptable salt thereof, wherein:
X is heteroaryl; wherein the heteroaryl represented by X is optionally substituted with one, two, three, or four R4;
Rx, at each occurrence, is independently selected from the group consisting of R5, OR5, SR5, S(O)R5, SO2R5, C(O)R5, CO(O)R5, OC(O)R5, OC(O)OR5, NH2, NHR5, N(R5)2, NHC(O)R5, NR5C(O)R5, NHS(O)2R5, NR5S(O)2R5, NHC(O)OR5, NR5C(O)OR5, NHC(O)NH2, NHC(O)NHR5, NHC(O)N(R5)2, NR5C(O)NHR5, NR5C(O)N(R5)2, C(O)NH2, C(O)NHR5, C(O)N(R5)2, C(O)NHOH, C(O)NHOR5, C(O)NHSO2R5, C(O)NR5SO2R5, SO2NH2, SO2NHR5, SO2N(R5)2, CO(O)H, C(O)H, OH, CN, N5, NO2, F, Cl, Br and I;
L1 is selected from the group consisting of (CR6R7)q, (CR6R7)s—O—(CR6R7)r, (CR6R7)s—C(O)—(CR6R7)r, (CR6R7)s—S—(CR6R7)r, (CR6R7)s—S(O)2—(CR6R7)r, (CR6R7)s—NR6AC(O)—(CR6R7)r, (CR6R7)s—C(O)NR6A—(CR6R7)r, (CR6R7)s—NR6A—(CR6R7)r, (CR6R7)s—S(O)2NR6A—(CR6R7)r, and (CR6R7)s—NR6AS(O)2—(CR6R7)r;
Y2 is C8-14 cycloalkyl, C8-14 cycloalkenyl, C8-14 heterocycloalkyl, or C8-14 heterocycloalkenyl; optionally fused to one or two rings selected from the group consisting of C3-8 cycloalkane, C3-8 cycloalkene, benzene, C5-6 heteroarene, C3-8 heterocycloalkane, and C3-8 heterocycloalkene; wherein Y2 is optionally substituted with one, two, three, four, or five substituents independently selected from the group consisting of R8, OR8, SR8, S(O)R8, SO2R8, C(O)R8, CO(O)R8, OC(O)R8, OC(O)OR8, NH2, NHR8, N(R8)2, NHC(O)R8, NR8C(O)R8, NHS(O)2R8, NR8S(O)2R8, NHC(O)OR8, NR8C(O)OR8, NHC(O)NH2, NHC(O)NHR8, NHC(O)N(R8)2, NR8C(O)NHR8, NR8C(O)N(R8)2, C(O)NH2, C(O)NHR8, C(O)N(R8)2, C(O)NHOH, C(O)NHOR8, C(O)NHSO2R8, C(O)NR8SO2R8, SO2NH2, SO2NHR8, SO2N(R8)2, CO(O)H, C(O)H, OH, CN, N3, NO2, F, Cl, Br and I;
Z1 is selected from the group consisting of C(O)OR9, C(O)NR10R11, C(O)R11, NR10C(O)R11, NR10C(O)NR10R11, OC(O)NR10R11, NR10C(O)OR9, C(═NOR10)NR10R11, NR10C(═NCN)NR10R11, NR10S(O)2NR10R11, S(O)2R9, S(O)2NR10R11, N(R10)S(O)2R11, NR10C(═NR11)NR10R11, C(═S)NR10R11, C(═NR10)NR10R11, halogen, NO2, and CN; or
Z1 is selected from the group consisting of
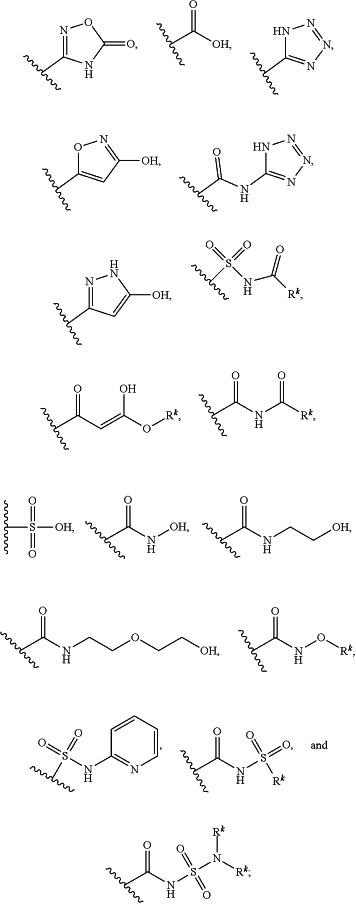 R1, at each occurrence, is independently selected from the group consisting of halo, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and C1-6 haloalkyl;
R2, at each occurrence, is independently selected from the group consisting of deuterium, halo, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and C1-6 haloalkyl;
two R2 that are attached to the same carbon atom, together with said carbon atom, optionally form a ring selected from the group consisting of heterocycloalkyl, heterocycloalkenyl, cycloalkyl, and cycloalkenyl;
R3, at each occurrence, is independently selected from the group consisting of halo, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and C1-6 haloalkyl;
R4, at each occurrence, is independently selected from the group consisting of NR12R13, OR12, CN, NO2, halogen, C(O)OR12, C(O)NR12R13, NR12C(O)R13, NR12S(O)2R14, NR12S(O)R14, S(O)2R14, S(O)R14 and R14;
R5, at each occurrence, is independently selected from the group consisting of C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C1-6 haloalkyl, C1-6 hydroxyalkyl, aryl, heterocyclyl, cycloalkyl, and cycloalkenyl;
R6A is independently selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and C1-6 haloalkyl;
R6 and R7, at each occurrence, are each independently selected from the group consisting of hydrogen, R15, OR15, SR15, S(O)R15, SO2R15, C(O)R15, CO(O)R15, OC(O)R15, OC(O)OR15, NH2, NHR15, N(R15)2, NHC(O)R15, NR15C(O)R15, NHS(O)2R15, NR15S(O)2R15, NHC(O)OR15, NR15C(O)OR15, NHC(O)NH2, NHC(O)NHR15, NHC(O)N(R15)2, NR15C(O)NHR15, NR15C(O)N(R15)2, C(O)NH2, C(O)NHR15, C(O)N(R15)2, C(O)NHOH, C(O)NHOR15, C(O)NHSO2R15, C(O)NR15SO2R15, SO2NH2, SO2NHR15, SO2N(R15)2, CO(O)H, C(O)H, OH, CN, N3, NO2, F, Cl, Br and I;
R8, at each occurrence, is independently selected from the group consisting of C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C1-6 haloalkyl, aryl, heterocyclyl, cycloalkyl, and cycloalkenyl; wherein the R8 C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and C1-6 haloalkyl are optionally substituted with one, two, three, four, five, or six substituents independently selected from the group consisting of R16, OR16, SR16, S(O)R16, SO2R16, C(O)R16, CO(O)R16, OC(O)R16, OC(O)OR16, NH2, NHR16, N(R16)2, NHC(O)R16, NR16C(O)R16, NHS(O)2R16, NR16S(O)2R16, NHC(O)OR16, NR16C(O)OR16, NHC(O)NH2, NHC(O)NHR16, NHC(O)N(R16)2, NR16C(O)NHR16, NR16C(O)N(R16)2, C(O)NH2, C(O)NHR16, C(O)N(R16)2, C(O)NHOH, C(O)NHOR16, C(O)NHSO2R16, C(O)NR16SO2R16, SO2NH2, SO2NHR16, SO2N(R16)2, CO(O)H, C(O)H, OH, CN, N3, NO2, F, Cl, Br and I; wherein the R8 aryl, heterocyclyl, cycloalkyl, and cycloalkenyl are optionally substituted with one, two, or three substituents independently selected from the group consisting of C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C1-6 haloalkyl, NH2, C(O)NH2, SO2NH2, C(O)H, (O), OH, CN, NO2, OCF3, OCF2CF3, F, Cl, Br and I;
R9 is selected from the group consisting of C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C1-6 haloalkyl, cycloalkyl, phenyl and (CH2)1-4 phenyl; and
R10 and R11, at each occurrence, are each independently selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl, C1-6 haloalkyl, phenyl and (CH2)1-4-phenyl; or
R10 and R11, or R10 and R9, together with the atom to which each is attached are combined to form a heterocyclyl;
Rk, at each occurrence, is independently selected from the group consisting of C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-7 heterocycloalkyl, C3-7 cycloalkyl and C1-6 haloalkyl;
R12 and R13, at each occurrence, are each independently selected from the group consisting of hydrogen, C1-4 alkyl, C2-4 alkenyl, C2-4 alkynyl, C1-4 haloalkyl and (CH2)1-4 phenyl;
R14, at each occurrence, is independently selected from the group consisting of C1-4 alkyl, C2-4 alkenyl, C2-4 alkynyl and C1-4 haloalkyl;
R12 and R13, or R12 and R14, at each occurrence, together with the atom to which each is attached, are optionally combined to form a heterocyclyl;
R15, at each occurrence, is independently selected from the group consisting of C1-4 alkyl, C2-4 alkenyl, C2-4 alkynyl, C1-4 haloalkyl, C1-4 hydroxyalkyl, aryl, heterocyclyl, cycloalkyl, and cycloalkenyl; wherein the R15 C1-4 alkyl, C2-4 alkenyl, C2-4 alkynyl, C1-4 haloalkyl, and C1-4 hydroxyalkyl are optionally substituted with one, two, or three substituents independently selected from the group consisting of O—(C1-4 alkyl), NH2, C(O)NH2, SO2NH2, C(O)H, C(O)OH, (O), OH, CN, NO2, OCF3, OCF2CF3, F, Cl, Br and I;
R16, at each occurrence, is independently selected from the group consisting of C1-4 alkyl, C2-4 alkenyl, C2-4 alkynyl, C1-4 haloalkyl, C1-4 hydroxyalkyl, aryl, heterocycloalkyl, heterocycloalkenyl, heteroaryl, cycloalkyl, and cycloalkenyl; wherein the R16 C1-4 alkyl, C2-4 alkenyl, C2-4 alkynyl, C1-4 haloalkyl, and C1-4 hydroxyalkyl are optionally substituted with one substituent independently selected from the group consisting of OCH3, OCH2CH2OCH3, and OCH2CH2NHCH3;
q is 1, 2, or 3;
s is 0, 1, or 2;
r is 0, 1, or 2;
wherein the sum of s and r is 0, 1, or 2;
m is 0, 1, 2, or 3;
n is 0, 1, 2, 3, 4, 5, or 6;
o is 0, 1, 2, 3, or 4; and
p is 0, 1, or 2.
|