| CPC A61K 31/16 (2013.01) [A61K 9/0065 (2013.01); A61K 9/0095 (2013.01); A61K 9/2846 (2013.01); A61K 9/2866 (2013.01); A61K 9/5073 (2013.01); A61K 9/5084 (2013.01); A61K 31/145 (2013.01); A61K 31/6615 (2013.01); C07C 323/25 (2013.01); C07C 323/52 (2013.01); C07F 9/65616 (2013.01); G01N 33/5023 (2013.01)] | 20 Claims |
|
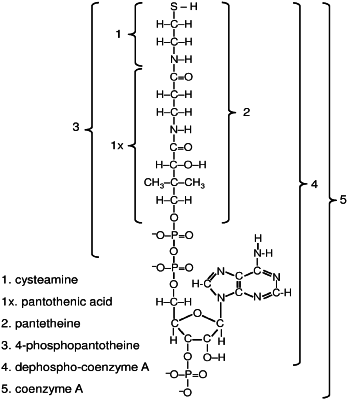
1. A method of treating a cysteamine sensitive disorder in a subject in need thereof, said method comprising administering to the subject a therapeutically-effective amount of a compound selected from pantetheine-N-acetyl-L-cysteine disulfide, pantetheine-N-acetylcysteamine disulfide, cysteamine-pantetheine disulfide, cysteamine-4-phosphopantetheine disulfide, cysteamine-gamma-L-glutamyl-L-cysteine disulfide, and cysteamine-N-acetyl-L-cysteine disulfide, and salts thereof to treat the cysteamine sensitive disorder,
wherein the cysteamine sensitive disorder is selected from Huntington's disease, Parkinson's disease, cystinosis; sickle cell disease; chronic obstructive pulmonary disease (COPD), cystic fibrosis (CF), non-alcoholic steatohepatitis (NASH), alcoholic steatohepatitis, non-alcoholic fatty liver disease (NAFLD), Rett syndrome, and mitochondrial encephalomyopathy lactic acidosis and stroke-like episodes (MELAS).
|