| CPC A61M 25/002 (2013.01) [A61B 50/20 (2016.02); A61B 50/30 (2016.02); A61B 50/33 (2016.02); A61L 2/0088 (2013.01); A61B 18/1492 (2013.01); A61B 2050/005 (2016.02); A61B 2050/3008 (2016.02); A61B 2050/3015 (2016.02); A61B 2050/314 (2016.02)] | 12 Claims |
|
1. A packaging system for storing a deployable medical device within a sterilized environment, the packaging system comprising:
a sealed container with at least one sealed seam;
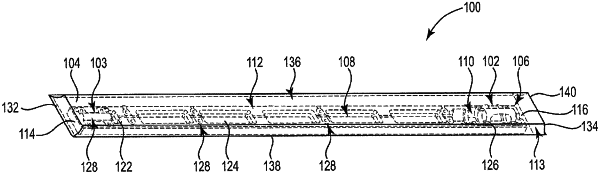
a tray disposed within the sealed container, the tray having a first end, a second end, a top wall, and a bottom wall, the tray comprising:
a first chamber near the first end;
a second chamber near the second end in fluid communication with the first chamber;
at least a third chamber positioned between the first chamber and the second chamber, the third chamber in fluid communication with the first chamber and the second chamber;
a first rib portion in the top wall between the first chamber and the third chamber, the first rib portion comprising a first channel extending between the first chamber and the third chamber;
a fourth chamber positioned between the second chamber and the third chamber, the fourth chamber in fluid communication with the first chamber, the second chamber, and the third chamber; and
a second rib portion in the top wall between the second chamber and the fourth chamber, the second rib portion comprising a second channel extending between the second chamber and the fourth chamber;
a catheter assembly having a distal end and a proximal end, the catheter assembly comprising at least one sheath and spanning between the first chamber and the second chamber;
a deployable medical device comprising a tissue material, the deployable medical device engaged with the catheter assembly and disposed within the tray in a storage configuration, wherein in the storage configuration, the deployable medical device is disposed within the first chamber in a partially deployed state distally from a distal end of the catheter assembly; and
a sterilizing fluid disposed within at least the first chamber, wherein the sterilizing fluid contacts at least a part of the deployable medical device in the storage configuration, and wherein each of the first chamber, second chamber, third chamber, and fourth chamber contain a volume of the sterilizing fluid.
|