| CPC A61L 33/0011 (2013.01) [A61K 9/0048 (2013.01); A61K 31/727 (2013.01); A61K 38/1709 (2013.01); A61K 47/55 (2017.08); A61K 47/61 (2017.08); A61K 47/64 (2017.08); A61L 27/54 (2013.01); A61L 29/16 (2013.01); A61L 31/16 (2013.01); A61P 7/02 (2018.01); A61P 27/02 (2018.01); A61L 2300/42 (2013.01); A61L 2420/02 (2013.01)] | 19 Claims |
|
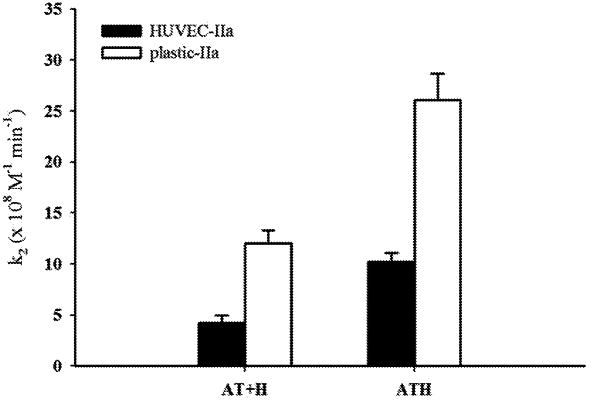
1. A medical device having reduced thrombogenicity, the medical device comprising a polymeric surface having a compound bonded to the polymeric surface of the medical device, wherein the compound includes an antithrombin-heparin conjugate,
wherein at least 98% of heparin chains in the antithrombin-heparin conjugate have a molecular weight greater than 5,000 Daltons,
wherein the antithrombin-heperain conjugate is conjugated at the aldose terminus aldehyde on the heparin and a lysyl or N-terminal amino group on antithrombin.
|