|
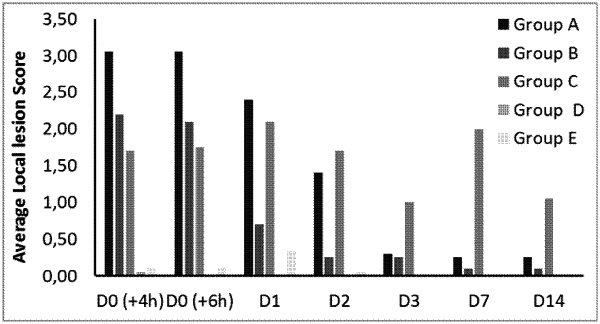
1. A method for the prevention and/or treatment of porcine enzootic pneumonia and/or Porcine Circovirus-Associated Diseases (PCVAD) comprising administering into the dermis of livestock in need thereof a single-dose combination vaccine comprising one or more antigens of Mycoplasma hyopneumoniae, one or more antigens of porcine circovirus, an adjuvant and pharmaceutically acceptable excipients and/or carriers, wherein the one or more antigens of porcine circovirus comprises the PCV2 ORF2 protein in an amount from 0.1 μg/dose to 10 μg/dose.
|