| CPC A61K 35/612 (2013.01) [A61K 9/0019 (2013.01); A61K 9/0053 (2013.01); A61K 31/047 (2013.01); A61K 31/122 (2013.01); A61K 31/685 (2013.01); A61P 27/02 (2018.01); C12P 9/00 (2013.01)] | 19 Claims |
|
1. A method for improving, controlling, reducing, or alleviating one or more symptoms and/or signs of an eye disease or condition in a subject in need thereof comprising:
administering to the subject in need thereof an effective amount of a pharmaceutical or nutraceutical formulation comprising a lysophosphatidylcholine (LPC) composition comprising a LPC-compound selected from the group consisting of any one of formulas 1 to 8, and any combination thereof, so that the symptoms of the disease or condition are improved, controlled, reduced, or alleviated:
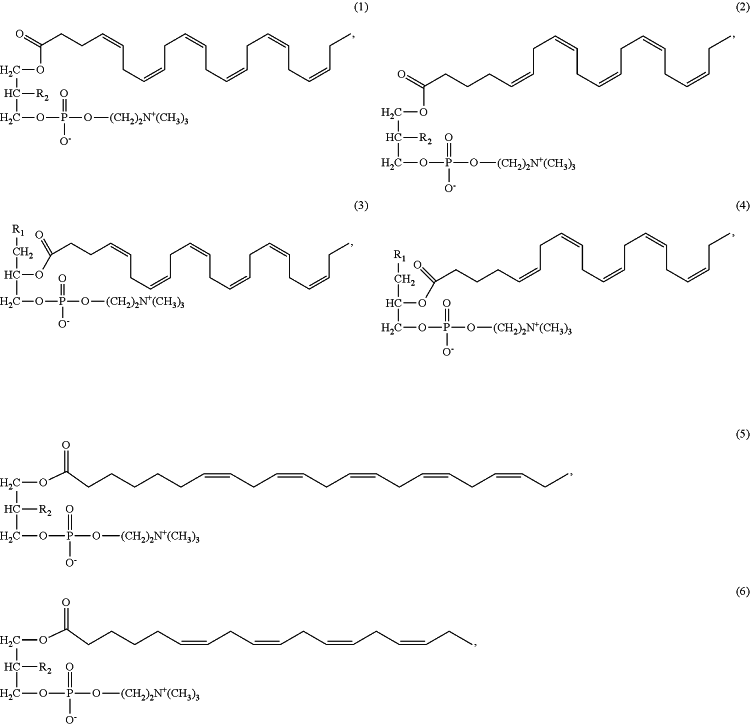 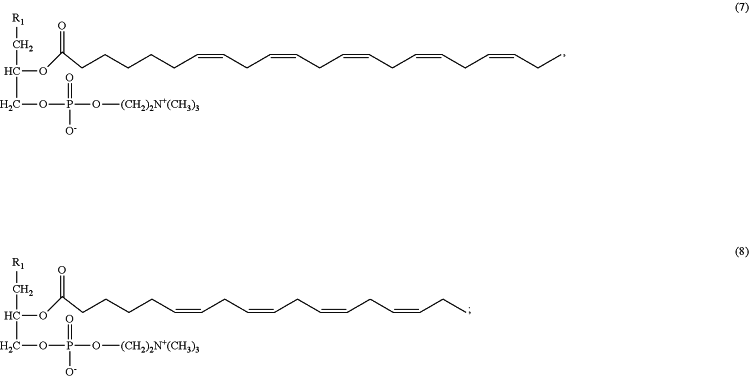 wherein:
R1 is OH or —OC(O)(CH2)nCH3;
R2 is OH or —OC(O)(CH2)nCH3; and
n is 0, 1, or 2;
and wherein the molar ratio of lysoPC-EPA:lysoPC-DHA is in the range of from 1:1 to 5:1; and further wherein
i) the number of moles of lysoPC-EPA is the number of moles 1-lysoPC-EPA plus the number of moles 2-lysoPC-EPA; and
ii) ii) the number of moles of lysoPC-DHA is the number of moles 1-lysoPC-DHA plus the number of moles 2-lysoPC-DHA.
|