| CPC A61M 1/3496 (2013.01) [A61M 1/3607 (2014.02); A61M 1/3616 (2014.02); A61M 1/3643 (2013.01); A61M 1/3672 (2013.01); A61M 1/38 (2013.01); A61M 1/382 (2013.01); A61M 1/385 (2013.01); G01N 33/48 (2013.01); G16H 10/60 (2018.01); A61M 2202/0415 (2013.01); A61M 2205/3379 (2013.01); A61M 2205/3393 (2013.01); A61M 2205/502 (2013.01); A61M 2205/505 (2013.01); A61M 2230/207 (2013.01)] | 23 Claims |
|
1. A method of collecting plasma, comprising:
receiving input from an operator via a touchscreen of a plasma collection device;
receiving donor parameters at a controller of the plasma collection device, wherein the donor parameters are received electronically from a donor management system;
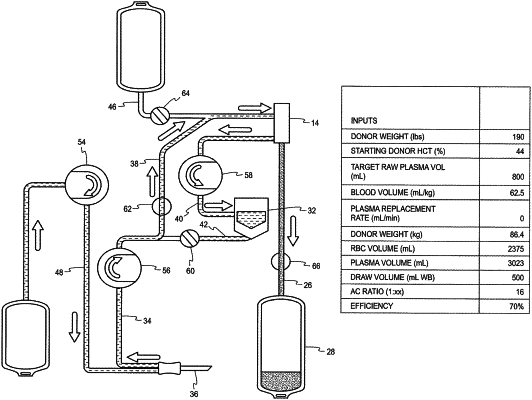
storing a target volume for raw plasma which is based at least in part on donor height and weight used to calculate total donor blood volume, the target volume for raw plasma based on the total donor blood volume;
setting the target volume for raw plasma prior to blood collection from a donor;
controlling the plasma collection device to operate draw and return phases to withdraw whole blood from the donor and separate the whole blood into a plasma product and a second blood component comprising red blood cells and to return the second blood component to the donor,
wherein each draw and return phase comprises:
introducing whole blood from the donor to a separator via a donor line;
combining anticoagulant with the whole blood from the donor using an anticoagulant line based on an anticoagulant ratio (ACR);
separating whole blood into the plasma product and the second blood component comprising red blood cells; and
sending the plasma product to a plasma product collection container,
wherein the controller is further configured to operate the draw and return phases until a volume of raw plasma (VRP) in the plasma product collection container equals the target volume of raw plasma, the volume of raw plasma (VRP) based on a measured volume of plasma product (VPP).
|