| CPC C07K 16/2809 (2013.01) [C07K 16/28 (2013.01); C07K 16/2878 (2013.01); A61K 2039/505 (2013.01); C07K 2317/31 (2013.01); C07K 2317/35 (2013.01); C07K 2317/52 (2013.01); C07K 2317/522 (2013.01); C07K 2317/55 (2013.01); C07K 2317/565 (2013.01); C07K 2317/73 (2013.01); C07K 2317/92 (2013.01)] | 28 Claims |
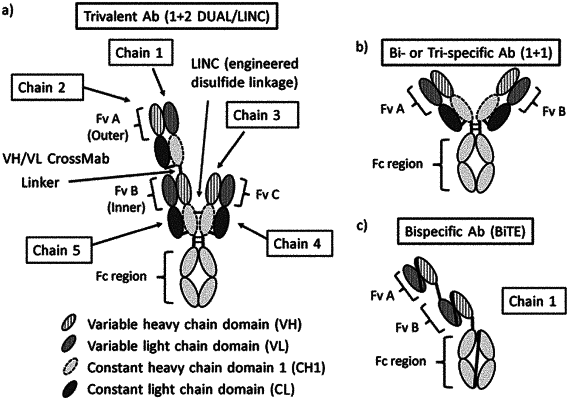
|
1. A pharmaceutical composition comprising
(1) a multispecific antigen-binding molecule that comprises:
(a) a first antigen-binding moiety and a second antigen-binding moiety, at least one of which binds to human CD137 and comprises an antibody variable region comprising a heavy chain CDR 1 comprising SEQ ID NO: 20, a heavy chain CDR 2 comprising SEQ ID NO: 34, a heavy chain CDR 3 comprising SEQ ID NO: 48, a light chain CDR 1 comprising SEQ ID NO: 63, a light chain CDR 2 comprising SEQ ID NO: 68, and a light chain CDR 3 comprising SEQ ID NO: 73; and
(b) a third antigen-binding moiety that binds to human Delta-like 3 (DLL3) and comprises an antibody variable region comprising a heavy chain CDR1 comprising SEQ ID NO: 233, a heavy chain CDR 2 comprising SEQ ID NO: 234, a heavy chain CDR 3 comprising SEQ ID NO: 235, a light chain CDR 1 comprising SEQ ID NO: 237, a light chain CDR 2 comprising SEQ ID NO: 238, and a light chain CDR 3 comprising SEQ ID NO: 239; and
(2) a pharmaceutically acceptable carrier.
|