| CPC A61K 31/519 (2013.01) [A61K 9/0019 (2013.01); A61K 9/19 (2013.01); A61K 31/4985 (2013.01); A61K 47/02 (2013.01)] | 11 Claims |
|
1. A method for preparing a lyophilized solid pharmaceutical composition comprising
a) a compound having the formula (I):
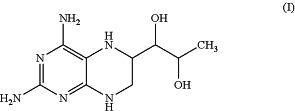 and/or a compound having the formula (II):
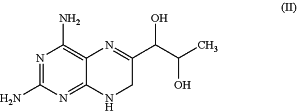 and
b) two different sodium phosphate salts, wherein the two different sodium phosphate salts are NaH2PO4·2H2O and Na2HPO4·2H2O, and wherein the quantity of the NaH2PO4·2H2O and Na2HPO4·2H2O present in the composition is chosen such that the molar ratio of both NaH2PO4·2H2O and Na2HPO4·2H2O to compound (I) or compound (II) ranges from 0.02 to 0.5 and optionally NaCl;
the method comprising:
aa) dissolving the compound of the formula (III) and/or (II):
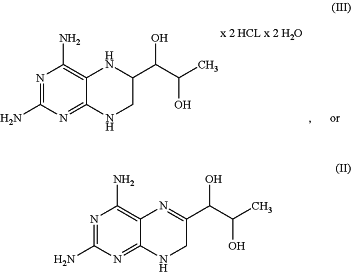 with a buffer,
wherein the buffer comprises the two different sodium phosphate salts, wherein the buffer is prepared by
separately dissolving in water NaH2PO4·2H2O and Na2HPO4·2H2O,
then adding the NaH2PO4·2H2O solution to the Na2HPO4·2H2O solution to obtain a sodium phosphate solution with a pH of 7.4, and
then adding the sodium phosphate solution to a 5N NaOH solution; and
bb) lyophilization of the solution obtained in aa).
|