| CPC C07K 16/2833 (2013.01) [A61K 47/6803 (2017.08); A61K 47/6811 (2017.08); A61K 47/6831 (2017.08); A61K 47/6867 (2017.08); A61P 35/00 (2018.01)] | 29 Claims |
|
1. An antibody conjugate comprising an antibody that specifically binds to CD74 linked site-specifically to at least one payload moiety, wherein the antibody comprises a non-natural amino acid at a site selected from the group consisting of HC-F404, HC-K121, HC-Y180, HC-F241, HC-221, LC-T22, LC-S7, LC-N152, LC-K42, LC-E161, LC-D170, HC-S136, HC-S25, HC-A40, HC-S119, HC-S190, HC-K222, HC-R19, HC-Y52, and HC-S70,
wherein the payload moiety is covalently linked to the non-natural amino acid directly or via a linker,
and wherein the antibody comprises three heavy chain CDRs and three light chain CDRs of a VH-VL pair selected from the group consisting of:
a. the VH region SEQ ID NO: 236 and the VL region SEQ ID NO: 256;
b. the VH region SEQ ID NO: 236 and the VL region SEQ ID NO: 257;
c. the VH region SEQ ID NO: 236 and the VL region SEQ ID NO: 265;
d. the VH region SEQ ID NO: 236 and the VL region SEQ ID NO: 264;
e. the VH region SEQ ID NO: 236 and the VL region SEQ ID NO: 267;
f. the VH region SEQ ID NO: 236 and the VL region SEQ ID NO: 268;
g. the VH region SEQ ID NO: 236 and the VL region SEQ ID NO: 269;
h. the VH region SEQ ID NO: 236 and the VL region SEQ ID NO: 270;
i. the VH region SEQ ID NO: 237 and the VL region SEQ ID NO: 256;
j. the VH region SEQ ID NO: 237 and the VL region SEQ ID NO: 257;
k. the VH region SEQ ID NO: 237 and the VL region SEQ ID NO: 265;
l. the VH region SEQ ID NO: 237 and the VL region SEQ ID NO: 264;
m. the VH region SEQ ID NO: 237 and the VL region SEQ ID NO: 267;
n. the VH region SEQ ID NO: 237 and the VL region SEQ ID NO: 268;
o. the VH region SEQ ID NO: 237 and the VL region SEQ ID NO: 269;
p. the VH region SEQ ID NO: 237 and the VL region SEQ ID NO: 270;
q. the VH region SEQ ID NO: 238 and the VL region SEQ ID NO: 256;
r. the VH region SEQ ID NO: 238 and the VL region SEQ ID NO: 257;
s. the VH region SEQ ID NO: 238 and the VL region SEQ ID NO: 265;
t. the VH region SEQ ID NO: 238 and the VL region SEQ ID NO: 264;
u. the VH region SEQ ID NO: 238 and the VL region SEQ ID NO: 267;
v. the VH region SEQ ID NO: 238 and the VL region SEQ ID NO: 268;
w. the VH region SEQ ID NO: 238 and the VL region SEQ ID NO: 269;
x. the VH region SEQ ID NO: 238 and the VL region SEQ ID NO: 270;
y. the VH region SEQ ID NO: 239 and the VL region SEQ ID NO: 256;
z. the VH region SEQ ID NO: 239 and the VL region SEQ ID NO: 257;
aa. the VH region SEQ ID NO: 239 and the VL region SEQ ID NO: 265;
bb. the VH region SEQ ID NO: 239 and the VL region SEQ ID NO: 264;
cc. the VH region SEQ ID NO: 239 and the VL region SEQ ID NO: 267;
dd. the VH region SEQ ID NO: 239 and the VL region SEQ ID NO: 268;
ee. the VH region SEQ ID NO: 239 and the VL region SEQ ID NO: 269;
ff. the VH region SEQ ID NO: 239 and the VL region SEQ ID NO: 270;
gg. the VH region SEQ ID NO: 235 and the VL region SEQ ID NO: 256;
hh. the VH region SEQ ID NO: 240 and the VL region SEQ ID NO: 256; and
ii. the VH region SEQ ID NO: 241 and the VL region SEQ ID NO: 256; and
wherein the conjugate is according to any of formulas 101a-104b:
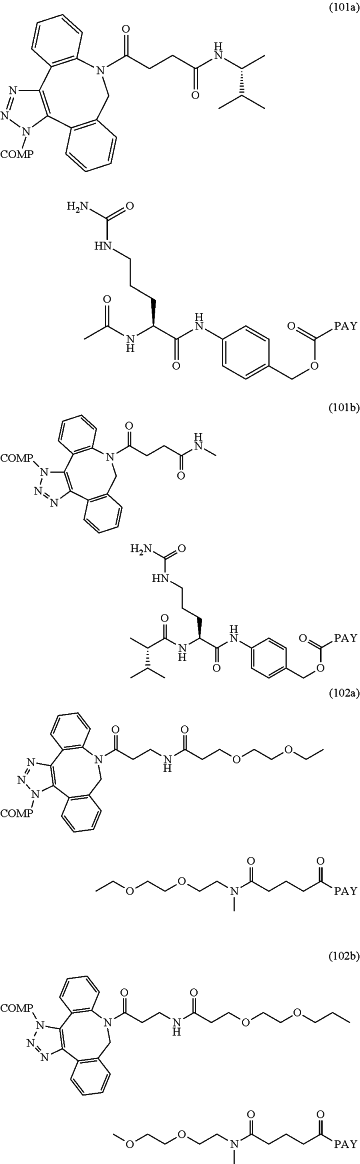 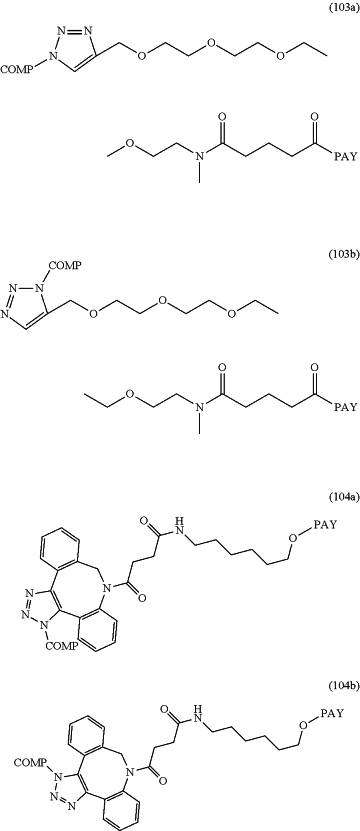 wherein COMP is the non-natural amino acid residue of the antibody and PAY is a payload moiety.
|