| CPC A61F 2/0045 (2013.01) [A61B 5/205 (2013.01); A61F 2/0027 (2013.01); A61B 17/3468 (2013.01); A61F 2230/006 (2013.01); A61F 2240/002 (2013.01); A61F 2250/0018 (2013.01); A61F 2250/0023 (2013.01); A61N 1/0514 (2013.01); A61N 1/36007 (2013.01)] | 13 Claims |
|
1. A subject-controlled implantable system for managing urinary incontinence, the system comprising:
a mesh-based urinary sling with an elongate body member having a proximal portion, a distal portion and an intermediate portion, wherein the intermediate portion of the elongate body member is configured to be positioned underneath a urethra of a subject for providing an adequate support to prevent leakage of urine during a stress event;
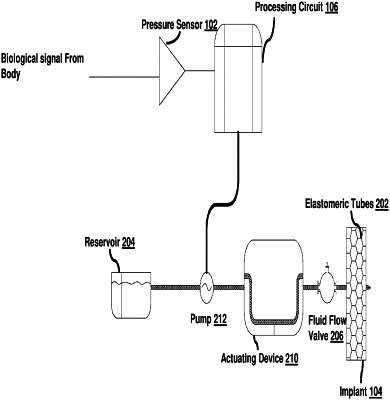
a trigger unit configured to be subcutaneously placed and configured to be activated manually by the subject upon a desire arising out of changing abdominal pressures transferring from an abdominal cavity, the trigger unit configured to generate a first signal upon activation by the subject;
a processing circuit to process the first signal, wherein the processing circuit is configured to generate a second signal to request an adjustment of a tensioning force in the elongate body member thereby changing a magnitude of a supporting force to the urethra; and
an elastomeric tube that is fabricated monolithically with the elongate body member, the tube including a lumen there-through for allowing circulation of a fluid, wherein the circulation of the fluid allows adjustments in the tensioning force in response to the second signal received from the processing circuit.
|