| CPC A61F 2/004 (2013.01) [A61F 2/04 (2013.01); A61B 2017/00805 (2013.01); A61F 2/0045 (2013.01); A61F 2002/047 (2013.01)] | 16 Claims |
|
1. An implantable device kit for controllable coaptation of a body lumen in tissue of a living body, comprising:
a sensor probe including a probe front end and a sensor incorporated onto the probe front end, the sensor is configured to detect information indicative of a degree of the coaptation of the body lumen; and
an implantable device including:
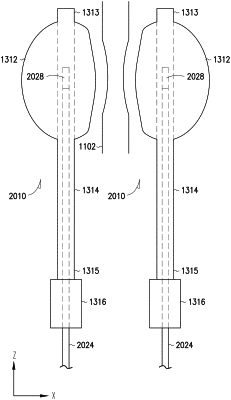
an adjustable membrane element including a continuous wall having an inner surface defining a chamber, the adjustable membrane element configured to coapt the body lumen;
an elongate conduit including a conduit peripheral surface, a conduit rear end, a conduit front end, a first conduit lumen, and a second conduit lumen, the conduit peripheral surface connected to and sealed to the adjustable membrane element at or near the conduit front end, the first conduit lumen having a first opening at the conduit rear end and a second opening in fluid communication with the chamber, the second conduit lumen configured to receive a portion of the sensor probe including the probe front end and to allow the probe front end to advance to a position at or near the conduit front end for the sensor to receive a signal including the information to be detected; and
a rear port connected to the conduit rear end and including a cavity in fluid communication with the first opening of the first conduit,
wherein the second conduit lumen comprises:
an outlet at or near the conduit front end allowing the probe front end including the sensor to protrude from the elongate conduit; and
a lumen shoulder,
wherein the sensor probe is configured for use as a push rod for advancing the implantable device in the tissue during an implantation of the implantable device and comprises a probe shoulder configured to abut the lumen shoulder to allow for transfer of force applied to the sensor probe to the implantable device.
|