| CPC A61K 31/4196 (2013.01) [A61K 31/437 (2013.01); A61K 31/4427 (2013.01); A61K 45/06 (2013.01); C07D 249/00 (2013.01); C07D 471/04 (2013.01)] | 13 Claims |
|
1. A compound of Formula (I)
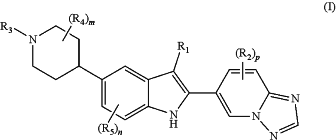 or a salt thereof, wherein:
R1 is H, Cl, —CN, C1-4 alkyl, C1-3 fluoroalkyl, C1-3 hydroxy-fluoroalkyl, —CRz═CH2, C3-6 cycloalkyl, —CH2(C3-6 cycloalkyl), —C(O)O(C1-3 alkyl), or tetrahydropyranyl;
each R2 is independently halo, —CN, —OH, —NO2+, C1-3 alkyl, C1-2 fluoroalkyl, C1-2 cyanoalkyl, C1-3 hydroxyalkyl, C1-3 aminoalkyl, —O(CH2)1-2OH, —(CH2)0-4O(C1-4 alkyl), C1-3 fluoroalkoxy, —(CH2)1-4O(C1-3 alkyl), —O(CH2)1-2OC(O)(C1-3 alkyl), —O(CH2)1-2NRxRx, —C(O)O(C1-3 alkyl), —C(O)NRyRy, —NRyRy, —NRy(C1-3 fluoroalkyl), —NRy(C1-4 hydroxyalkyl), —NRxCH2(phenyl), —NRxS(O)2(C3-6 cycloalkyl), —NRxC(O)(C1-3 alkyl), —NRx(CH2-cyclopropyl), C3-6 cycloalkyl, morpholinyl, dioxothiomorpholinyl, methylpiperidinyl, methylpiperazinyl, amino-oxadiazolyl, imidazolyl, triazolyl, or —C(O)(thiazolyl);
R3 is -L1-A;
L1 is —C(O)CH2— or —C(O)(CH2)2—;
A is 2-oxa-6-azaspiro[3,3]heptanyl, 4-oxaspiro[2.5]octanyl, 7-azaspiro[3.5]nonanyl, 8-azabicyclo[3.2.1]octanyl, 8-oxa-3-azabicyclo[3.2.1]octanyl, 9-azabicyclo[3.3.1]nonanyl, adamantanyl, azepanyl, azetidinyl, C3-6 cycloalkyl, diazepanyl, dihydroinonyl, dihydropyrimidinonyl, dioxanyl, dioxidothiadiazinanyl, dioxidothiazolidinyl, dioxidothiomorpholinyl, dioxoisothiazolidinyl, dioxidothiazinanyl, dioxotetrahydrothiophenyl, dioxotetrahydrothiopyranyl, dioxothiomorpholinyl, furanyl, imidazolyl, imidazolidinonyl, indolyl, isoquinolinyl, isoxazolyl, morpholinyl, morpholinonyl, naphthalenyl, octahydrocyclopenta[b]pyranyl, octahydropyrrolo[3,4-b]pyridinyl, oxazolidinonyl, oxadiazolyl, oxazolyl, oxetanyl, phenyl, piperidinyl, piperidinonyl, piperazinyl, piperazinonyl, pyrazinyl, pyrazolyl, pyridazinyl, pyridazinonyl, pyridinonyl, pyridinyl, pyrimidinyl, pyrrolidinonyl, pyrrolidinyl, pyrrolyl, quinolinyl, quinolizinonyl, tetrahydrofuranyl, tetrahydropyranyl, tetrahydrothiopyranyl, tetrazolyl, thiadiazolyl, thiazolyl, triazolonyl, or triazolyl, each substituted with -L2-Ra and zero to 4 Rb;
L2 is a bond or —CRxRx—;
Ra is:
(a) H, F, Cl, —CN, —OH, C1-6 alkyl, C1-3 fluoroalkyl, C1-5 hydroxyalkyl, —(CH2)0-4O(C1-3 alkyl), —(CRxRx)1-3S(C1-3 alkyl), —NHC(O)OC(CH3)3, —(CRxRx)1-3NHC(O)O(C1-4 alkyl), —(CRxRx)1-3NRyRy, —(CRxRx)1-3C(O)NRyRy, —O(C1-3 fluoroalkyl), —S(O)2NRxRx, —O(CRxRx)1-3NRxRx, —NHS(O)2(C1-3 alkyl), —NRxRx, —NRx(C1-4 alkyl), —NRxC(O)(C1-4 alkyl), —(CRxRx)0-3C(O)OH, —C(O)(C1-5 alkyl), —C(O)(C1-3 fluoroalkyl), —C(O)O(C1-4 alkyl), —C(O)NH(C1-3 cyanoalkyl), —C(O)NRyRy, —C(O)NRxCH2C(O)NRxRx, or —C(O)NRxCH2CH2NHC(O)(C1-3 alkyl);
(b) C3-6 cycloalkyl or —C(O)NH(C3-6 cycloalkyl), wherein each cycloalkyl is substituted with zero to 2 substituents independently selected from —OH, C1-3 alkyl, C1-3 hydroxyalkyl, C1-3 fluoroalkyl, and —C(O)O(C1-3 alkyl); or
(c) A1, —CH2A1, —C(O)A1, —NRxA1, or —C(O)NRxA1, wherein A1 is furanyl, imidazolyl, indolyl, isoxazolyl, morpholinyl, octahydropyrrolo[3,4-c]pyrrolyl, oxazolyl, oxetanyl, phenyl, piperazinonyl, piperazinyl, piperidinyl, pyrazinyl, pyrazolyl, pyridinyl, pyrimidinyl, pyrrolidinonyl, pyrrolidinyl, pyrrolyl, tetrahydrofuranyl, tetrahydropyranyl, thiadiazolyl, thiazolyl, thiophenyl, or triazolyl, each substituted with zero to three substituents independently selected from —OH, C1-3 alkyl, C1-3 hydroxyalkyl, —C(O)(C1-2 alkyl), —C(O)O(C1-3 alkyl), —NRxRx, phenyl, trifluoromethyl-phenyl, —CH2(bromophenyl), and —CH2CH2(pyrrolidinyl);
each Rb is independently F, —OH, —CH3, —CF3, or —OCH3;
each Rx is independently H or —CH3;
each Ry is independently H or C1-6 alkyl;
Rz is H, C1-2 alkyl, or C1-2 fluoroalkyl;
each R4 is independently F, —OH, C1-2 alkyl, or —OCH3; or two R4 attached to the same carbon atom form ═O; or wherein when m is at least 2, two R4, each attached to a different carbon atom adjacent to the nitrogen atom in the piperidinyl ring, can form a —CH2CH2— bridge;
each R5 is independently F, Cl, —CN, C1-2 alkyl, C1-2 fluoroalkyl, or —OCH3;
m is zero, 1, 2, 3, or 4;
n is zero, 1, or 2; and
p is zero, 1, 2, 3, or 4.
|