| CPC C12M 37/06 (2013.01) [A61L 2/28 (2013.01); C12M 23/38 (2013.01); C12Q 1/22 (2013.01)] | 18 Claims |
|
1. A method of determining the accuracy of a sterilization or decontamination process in a sterilization chamber comprising the steps of:
placing a self-contained biological indicator (SCBI) in said sterilization chamber in an open position wherein a sterilant gas will enter said SCBI;
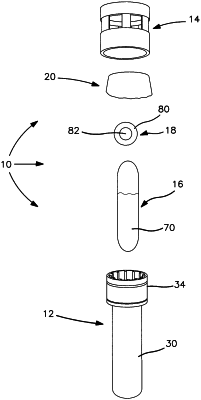
said SCBI comprising a body adapted to serve as a culture tube, said body having a cylindrical side wall which does not contain any recessed areas, a bottom wall at one end of the cylindrical side wall closing the body, a collar at the other end of the cylindrical side wall extending outwardly from the cylindrical side wall having an open end having an interior diameter greater than an interior diameter of the cylindrical side wall and adapted to receive a cap, said collar including a plurality of inwardly extending ribs; a cap adapted to engage said body in an open position and a closed position, said cap including a top wall and a cylindrical side wall adapted to engage said collar of said body, said cylindrical side wall including a plurality of apertures; a rupturable glass ampoule including a growth media in said body; and a stainless steel disc inoculated with spores on top of said glass ampoule and adjacent said plurality of ribs;
subjecting said sterilization chamber to said sterilant gas;
removing said SCBI from said sterilization chamber;
rupturing said glass ampoule in said SCBI to release said growth media to said body of said SCBI, wherein said stainless steel disc is adapted to drop down into said cylindrical side wall of said body to contact said inoculated spores with said growth media; and
closing said SCBI and determining the accuracy of said sterilization process or said decontamination process.
|