| CPC A61F 2/28 (2013.01) [A61F 2002/2828 (2013.01); A61F 2002/2835 (2013.01); A61F 2310/00179 (2013.01); A61F 2310/00796 (2013.01)] | 19 Claims |
|
1. An engineered medical osteonecrosis treatment device, comprising:
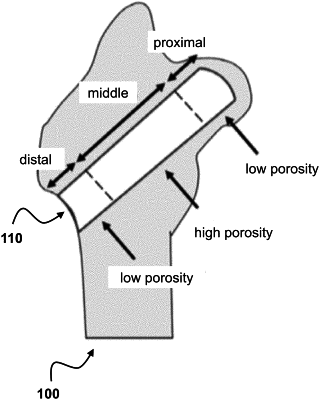
a cylindrically-shaped biodegradable scaffold having a central channel in a cylindrical alignment, wherein the central channel is sufficiently wide in diameter to contain cargo, wherein the cylindrically-shaped biodegradable scaffold is made of filaments having three cylindrical sections of spatially graded chemical composition, porosity and mechanical strength,
wherein the three cylindrical sections distinguish in the cylindrical alignment with each other a proximal cylindrical section, a distal cylindrical section and a single intermediary cylindrical section positioned in between the proximal and distal cylindrical sections,
wherein the intermediary cylindrical section is longer than each of the proximal and distal cylindrical sections,
wherein a porosity of the proximal and distal cylindrical sections across the entire proximal and distal cylindrical sections is less than a porosity across the entire intermediary cylindrical section, and wherein the proximal cylindrical section has a porosity which is less than the porosity of the distal cylindrical section, and wherein a mechanical strength across the entire proximal and distal cylindrical sections is higher than a mechanical strength across the entire intermediary cylindrical section.
|