| CPC C07K 16/244 (2013.01) [A61K 39/395 (2013.01); A61K 47/6845 (2017.08); C07K 16/2809 (2013.01); C07K 16/30 (2013.01); C07K 2317/31 (2013.01); C07K 2317/35 (2013.01); C07K 2317/52 (2013.01); C07K 2317/522 (2013.01); C07K 2317/53 (2013.01); C07K 2317/55 (2013.01); C07K 2317/76 (2013.01); C07K 2317/92 (2013.01); C07K 2317/94 (2013.01)] | 24 Claims |
|
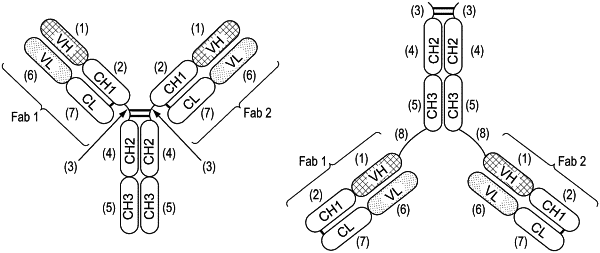
1. A pharmaceutical composition comprising an excipient and an antigen-binding molecule, the antigen-binding molecule comprising:
(a) a first polypeptide comprising, in an N- to C-terminal orientation:
(i) a first Fc domain;
(ii) a first linker;
(iii) a first VH domain; and
(iv) a first CH1 domain;
(b) a second polypeptide comprising, in an N- to C-terminal orientation:
(i) a second Fc domain;
(ii) a second linker;
(iii) a second VH domain; and
(iv) a second CH1 domain;
(c) a third polypeptide associated with the first polypeptide comprising, in an N-to-C terminal orientation, a first light chain comprising a first VL domain and a first CL domain, wherein the first VL domain and the first CL domain are associated with the first VH domain and the first CH1 domain to form a first Fab domain; and
(d) a fourth polypeptide associated with the second polypeptide comprising, in an N-to-C terminal orientation, a second light chain comprising a second VL domain and a second CL domain, wherein the second VL domain and the second CL domain are associated with the second VH domain and the second CH1 domain to form a second Fab domain that is different from the first Fab domain and binds to the same protein as the first Fab domain,
wherein the first light chain and the second light chain are universal light chains.
|