| CPC A61K 31/336 (2013.01) [A61J 1/035 (2013.01); A61K 31/194 (2013.01); A61K 31/215 (2013.01); A61K 31/225 (2013.01); A61K 31/343 (2013.01); A61K 31/381 (2013.01); A61K 31/403 (2013.01); A61K 9/4825 (2013.01)] | 7 Claims |
|
1. A dosage form for treating multiple sclerosis, comprising:
(a) a therapeutically effective amount of dimethyl fumarate in solid form;
(b) one or more compounds of Formula (IV):
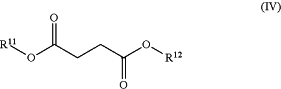 isomers thereof and salts of any of the foregoing, wherein:
each of R11 and R12 is independently chosen from hydrogen and methyl; and
wherein the one or more compounds of Formula (IV) are impurities and are present in a combined total amount of about 0.001% to 2% by weight based on total weight of the dimethyl fumarate; and
(c) a pharmaceutically acceptable vehicle.
|