| CPC A61B 5/7278 (2013.01) [A61B 5/08 (2013.01); A61B 5/25 (2021.01); A61B 7/003 (2013.01); A61B 7/04 (2013.01); A61N 1/046 (2013.01); A61N 1/0484 (2013.01); A61N 1/36542 (2013.01); A61N 1/3655 (2013.01); A61N 1/37247 (2013.01); A61N 1/37258 (2013.01); A61N 1/37282 (2013.01); A61N 1/3904 (2017.08); A61N 1/3993 (2013.01); A61B 5/363 (2021.01); A61B 5/4818 (2013.01); A61B 5/6805 (2013.01); A61B 5/6823 (2013.01); A61B 5/6844 (2013.01); A61B 5/721 (2013.01); A61B 2562/0219 (2013.01); A61B 2562/17 (2017.08); A61N 1/39044 (2017.08)] | 20 Claims |
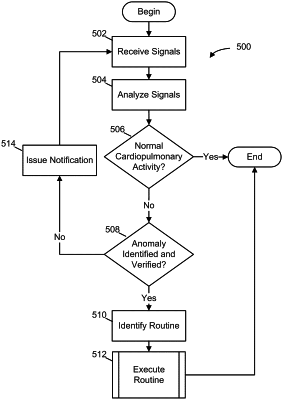
|
1. An ambulatory cardiac monitoring device configured to determine whether a patient is experiencing ventricular tachycardia (VT), the ambulatory cardiac monitoring device comprising:
electrocardiogram (ECG) electrodes configured to couple to the patient for an extended period of time;
an acoustic sensor; and
at least one processor coupled with the acoustic sensor and the ECG electrodes and configured to
receive at least one electrical signal from the ECG electrodes,
detect a pattern within the at least one electrical signal that is consistent with the patient experiencing VT,
receive at least one signal from the acoustic sensor,
determine whether the patient is experiencing VT based on the at least one signal from the acoustic sensor, and
in response to determining that the patient is experiencing VT based on the at least one signal from the acoustic sensor, record the at least one electrical signal and the at least one signal from the acoustic sensor.
|