| CPC C01F 11/24 (2013.01) [C02F 1/58 (2013.01); C02F 1/705 (2013.01); C08L 5/08 (2013.01); B82Y 30/00 (2013.01); B82Y 40/00 (2013.01); C01P 2004/64 (2013.01); C02F 2101/308 (2013.01); C02F 2103/30 (2013.01)] | 16 Claims |
|
1. A method, consisting of;
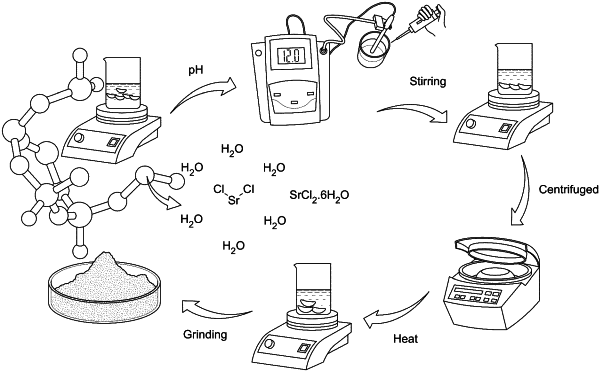
dissolving a strontium chloride hexahydrate in water and stirring the solution for 2 hours to make a dissolved solution of the strontium chloride hexahydrate;
adjusting a pH of the dissolved solution of strontium chloride hexahydrate to a specific pH by adding sodium hydroxide solution and stirring the pH adjusted sodium hydroxide solution for 2 hours to form a precipitate of a strontium chloride hexahydrate nanoparticle in solution;
centrifuging the a strontium chloride hexahydrate nanoparticle in solution to collect a pellet of the precipitate of the strontium chloride hexahydrate nanoparticle;
heating the pellet of the strontium chloride hexahydrate nanoparticle at 120° C. for 24 hours and grinding the heated pellet to obtain a powder of strontium chloride hexahydrate nanoparticle;
preparing a strontium chloride hexahydrate nanoparticle solution using the powder of strontium chloride hexahydrate nanoparticle in distilled water and adding a specific concentrations of a chitosan solution to create a chitosan (CS)-doped Strontium oxide (SrO) nanocomposite; and
reducing a pollutant content in water by adding the CS-doped SrO nanocomposite in presence of a reducing agent at a second specific pH to purify water.
|