| CPC A61K 47/541 (2017.08) [A61K 31/202 (2013.01); A61K 31/232 (2013.01); A61K 38/03 (2013.01); A61K 45/06 (2013.01); A61K 47/62 (2017.08); A61P 1/00 (2018.01); A61P 1/04 (2018.01); A61P 1/06 (2018.01); A61P 1/12 (2018.01); A61P 29/00 (2018.01); C07C 59/42 (2013.01); C07C 229/26 (2013.01); C07C 237/12 (2013.01); C07C 237/22 (2013.01); A61K 2800/00 (2013.01)] | 14 Claims |
|
1. A method for treating colon cancer in a subject in need of such treatment, the method comprising administering to the subject a pharmaceutical composition comprising a compound of Formula IV, or an enantiomer, polymorph, solvate, or hydrate thereof, and a carrier:
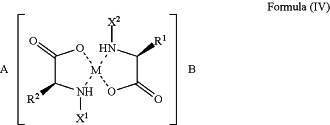 wherein
M is a divalent metal selected from magnesium (Mg2+), calcium (Ca2+), and zinc (Zn2+),
A and B are each an SPM molecule selected from an E series resolvin, a D series resolvin, and their aspirin-triggered counterparts,
R1 and R2 are each independently —(CH2)3—Y1, and —(CH2)4—Y2,
where Y1 and Y2 are each a basic function selected from a positively charged primary amine, a positively charged secondary amine, a positively charged tertiary amine, and a positively charged guanidine,
X1 and X2 are each independently H or CO—Z and Z is a peptide comprising 1 to 5 amino acids.
|