| CPC A61K 39/001169 (2018.08) [A61K 39/092 (2013.01); A61K 39/095 (2013.01); A61K 39/145 (2013.01); A61K 39/215 (2013.01); A61K 39/385 (2013.01); A61K 39/39 (2013.01); A61K 39/00117 (2018.08); A61K 39/001172 (2018.08); A61K 2039/575 (2013.01); A61K 2039/6031 (2013.01); A61K 2039/6037 (2013.01); A61K 2039/6056 (2013.01); A61K 2039/627 (2013.01); A61K 2039/70 (2013.01)] | 14 Claims |
|
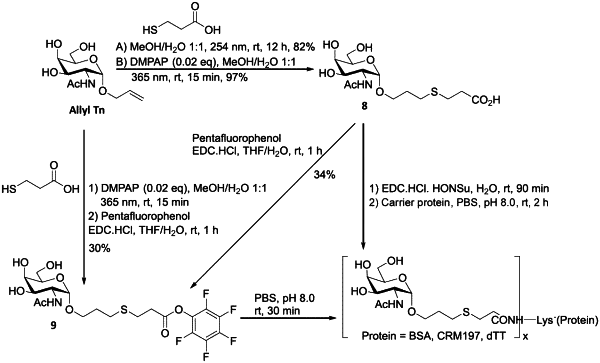
1. A vaccine composition comprising a synthetic neoglycoconjugate and an adjuvant, wherein the synthetic neoglycoconjugate comprises one or more carbohydrate antigens (CA) conjugated to one or more amine groups of an amino acid of a carrier protein or peptide (CP-NH) via a linker, the synthetic neoglycoconjugate having the structure:
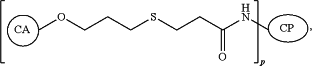 wherein p is an integer corresponding to the total number of carbohydrate antigens conjugated to the carrier protein or peptide at said one or more amine groups,
wherein the one or more carbohydrate antigens comprises unsialylated TF antigen, unsialylated Tn antigen, or a combination thereof,
wherein the carrier protein or peptide is, is from, or comprises Tetanus Toxoid, cross-reacting material 197, or an immunogenic fragment thereof, and
wherein administration of said vaccine composition triggers the production of antibodies against unsialylated TF antigen, unsialylated Tn antigen, or both unsialylated TF and unsialylated Tn antigens.
|