| CPC A61K 9/1694 (2013.01) [A61K 9/0046 (2013.01); A61K 31/47 (2013.01); A61K 47/02 (2013.01); A61K 47/26 (2013.01); A61K 47/38 (2013.01)] | 17 Claims |
|
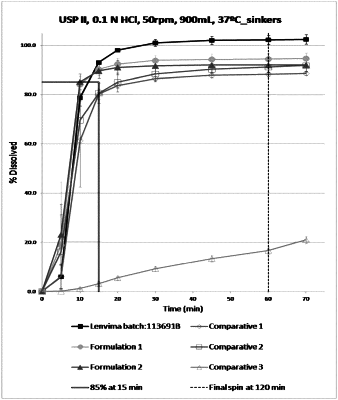
1. A pharmaceutical composition comprising (i) a therapeutically effective dose of Lenvatinib mesylate and (ii) sodium bicarbonate in a range of from 20 to 55%, by weight based on the total weight of the composition, wherein the weight ratio of Lenvatinib mesylate to sodium bicarbonate ranges from 1:1.5 to 1:5.
|