| CPC A61M 25/10 (2013.01) [A61B 17/12022 (2013.01); A61B 17/12109 (2013.01); A61B 17/12131 (2013.01); A61M 5/00 (2013.01); A61B 17/1214 (2013.01); A61B 17/12145 (2013.01); A61B 17/12159 (2013.01); A61B 17/12181 (2013.01); A61B 2017/00004 (2013.01); A61B 2017/00778 (2013.01); A61B 2017/00884 (2013.01); A61B 2017/00893 (2013.01); A61M 2025/1004 (2013.01); A61M 2025/105 (2013.01); A61M 2025/1065 (2013.01)] | 16 Claims |
|
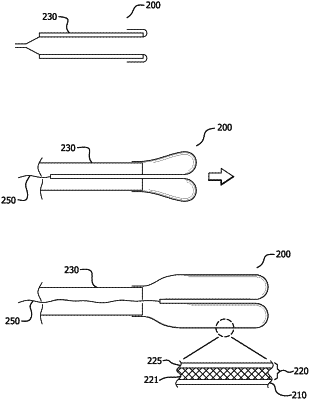
1. A vascular delivery device comprising:
an inner expandable member;
a drug infusible layer having a sleeve that circumscribes, is mounted around, or is situated about the inner expandable member defining at least a portion of interstitial space, the interstitial space at least partially containing a therapeutic agent, and the drug infusible layer being associated with the inner expandable member;
an outer barrier, wherein the outer barrier circumscribes or is mounted around the drug infusible layer; and
a tethering device comprising a guidewire, filament, or thread coupled to a distal end of the inner expandable member, the tethering device configured to:
(a) retract the inner expandable member and the drug infusible layer into a retracted position by pulling on the guidewire, filament, or thread to prevent the therapeutic agent from being transferred through the outer barrier; and
(b) extend the inner expandable member and the drug infusible layer into an extended position via pressurization to allow the therapeutic agent to transfer from the interstitial space of the drug infusible layer through the outer barrier in response to being compressed against an exterior vessel wall.
|