| CPC A61K 39/21 (2013.01) [C07K 1/18 (2013.01); C07K 14/005 (2013.01); C07K 14/162 (2013.01); C12N 7/02 (2013.01); A61K 2039/53 (2013.01); C12N 2740/16051 (2013.01); C12N 2740/16111 (2013.01); C12N 2740/16122 (2013.01); C12N 2740/16134 (2013.01)] | 14 Claims |
|
1. A process for purifying HIV-1 gp140 protein, comprising the steps of:
i) providing a composition comprising HIV-1 gp140 protein and other, non-desired host cell proteins derived from the host cell in which HIV-1 gp140 protein is expressed;
ii) capturing the HIV-1 gp140 protein on a multimodal resin comprising hydrophobic interaction and cation exchange properties, and eluting a purified fraction comprising the HIV-1 gp140 protein from the multimodal resin, wherein the multimodal resin comprising hydrophobic interaction and cation exchange properties comprises a solid support matrix with the following attached ligand of
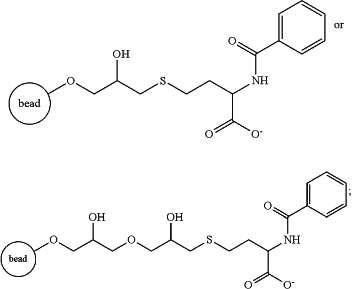 iii) applying the purified fraction of step ii) to an anion exchange resin to bind the HIV-1 gp140 protein, and eluting a further purified fraction comprising the HIV-1 gp140 protein from the anion exchange resin, wherein the anion exchange resin comprises a solid support matrix with a ligand of quaternary polyethyleneimine;
iv) applying the further purified fraction of HIV-1 gp140 protein of step iii) to an affinity medium resin comprising the ligand dextran sulfate, and eluting a further purified fraction comprising the HIV-1 gp140 protein from said resin; and,
v) subjecting the further purified fraction of step iii) to a multimodal resin that has anion-exchange and hydrophobic functionalities, wherein the multimodal resin that has anion-exchange and hydrophobic functionalities comprises a solid support matrix with a ligand of
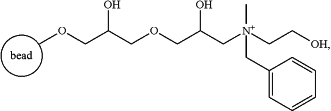 and eluting a further purified HIV-1 gp140 protein.
|